- Poly(ethylene glycol)-Keratin Hydrogels Prepared via Thiol-Maleimide Reaction
Department of Chemical Engineering, İzmir Institute of Technology, Urla, İzmir, 35430, Türkiye
- Thiol-Maleimide 반응을 이용한 Poly(ethylene glycol)-케라틴 하이드로젤 연구
Reproduction, stored in a retrieval system, or transmitted in any form of any part of this publication is permitted only by written permission from the Polymer Society of Korea.
The mechanical properties of hydrogels have a profound effect on cellular responses in tissue engineering applications. In this study, poly(ethylene glycol)-keratin (PEG-KRTN) hydrogels with tunable mechanical properties were prepared by varying molar mass of the maleimide functionalized PEG in the thiol-maleimide chemistry. Reduced keratins were reacted with PEG-maleimides having 2000 Da and 6000 Da molar masses. Viscoelastic and physiochemical properties and cytocompatibility of these hydrogels were tested. Storage modulus values were obtained as 2613 ± 254 Pa and 1313 ± 345 Pa for PEG2000-KRTN and PEG6000-KRTN hydrogels, respectively. Strain sweep data indicate that the linear viscoelastic region (LVER) of the PEG6000-KRTN hydrogel spans up to 40% strain value, whereas it is limited to 10% critical strain for the PEG2000-KRTN hydrogel. PEG6000-KRTN hydrogel presented higher swelling ratios and porosity. CCK-8 test showed that both hydrogels promoted the proliferation of L929 mouse fibroblast cells and, hence, can be applied in soft tissue engineering.
Reduced keratins were reacted with the PEG-dimaleimides (2000 Da or 6000 Da) to form self-standing hydrogels. Both hydrogels supported the growth of the fibroblast cells, confirming their biocompatibility.
Keywords: keratin, poly(ethylene glycol), michael addition reaction, hydrogel, rheology.
This work was funded by the İzmir Institute of Technology Research Foundation, grant number 2020IYTE0096. The Integrated Research Center at İzmir Institute of Technology is acknowledged for making XRD, SEM, and UV-Vis spectroscopy analyses possible.
The authors declare that there is no conflict of interest.
Hydrogels are exceptional scaffolds in tissue engineering applications as they are structurally similar to native tissues, and their biochemical and structural properties can be modulated to mimic extracellular matrix to maintain homeostasis.1,2
Incorporation of the cell adhesion ligands or degradable sequences into the hydrogel matrices has been widely exploited to impart bioactivity to the hydrogel matrices.3-6
Hydrogel mechanical properties, on the other hand, also influence the morphology, proliferation, migration, signaling, and differentiation of the cells.7-10
The mechanical properties of hydrogels can be tuned via chemical crosslinking methods. However, the crosslinking agents and byproducts generated can be toxic.11
Thus, biocompatible reagents and addition reactions should be used in the crosslinking reactions to overcome potential toxicity problems. The Michael-type addition reactions require a nucleophile as an electron donor and an electron-deficient carbon-carbon double bond present in functional groups such as acrylates, maleimides, and vinyl sulfones as an electron acceptor.12,13
Maleimides are the most efficient Michael-type acceptors used in the thiol-engaged Michael addition reactions, which ensure rapid crosslinking at physiological conditions.14-16
Maleimide functionalized poly(ethylene glycol) (PEG) has been frequently preferred in hydrogel formulations due to its biocompatibility, hydrophilicity, and biologically inertness.15,17
Especially, the hydrogels crosslinked by PEG-maleimides were proposed in cell encapsulation applications where in situ gelation is required.15,18,19
Keratin, a fibrous protein extracted from natural sources such as hair, nails, and wool with inherent biocompatibility and biodegradability, is a sustainable hydrogel-forming biopolymer that can serve as a tissue engineering scaffold and drug delivery platform.20,21
Mechanical and chemical stabilities of the keratin are mainly governed by the disulfide linkages in its structure. However, the reduction of keratins enhances their solubility in water by generating free thiols, which can be exploited in various functionalization and crosslinking processes.22,23
Additionally, the presence of bioactive domains such as LDV (Leu-Asp-Val), EDS (Glu-Asp-Ser), and RGD (Arg- Gly-Asp) in the keratin proteins promotes the cell attachment characteristics of the keratinous hydrogel matrices.24,25
In this study, chemically crosslinked PEG-keratin hydrogels were prepared via a thiol-maleimide reaction (Scheme 1). Reduced keratins were used as a Michael-type donor source and reacted with homobifunctional PEG-maleimides (6000 Da or 2000 Da) to form biocompatible hydrogels with tunable viscoelastic and physicochemical properties.
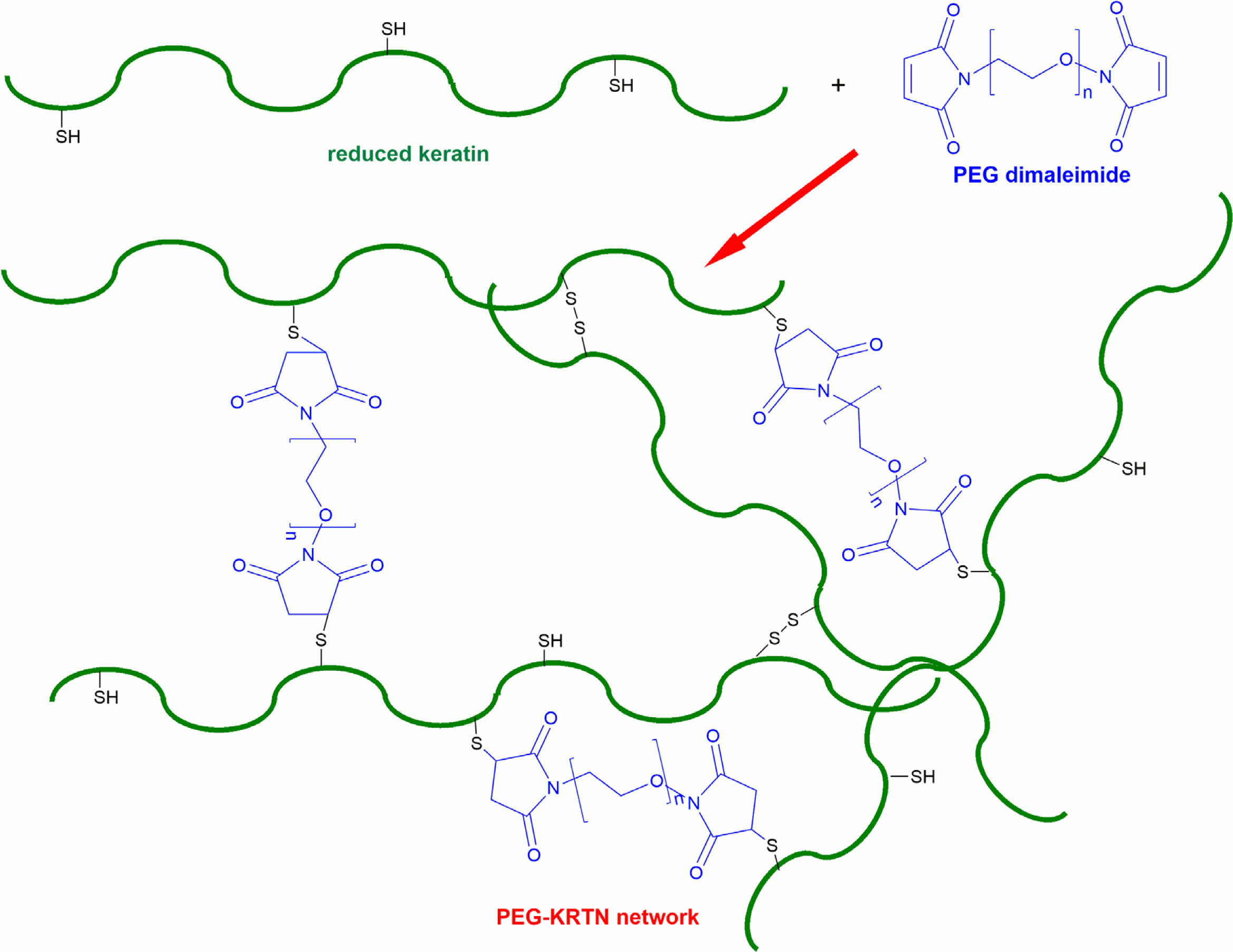
Scheme 1. The network structure of PEG-KRTN prepared via thiol-maleimide reaction and disulfide bond formation.
Materials. Karacabey Merino sheep wool was kindly provided by the Sheep Breeding Research Institute (Balıkesir, Turkey). Chloroform, methanol, sodium sulfide nonahydrate, DL-dithiothreitol (DTT), 5,5’-dithio-bis-(2-nitrobenzoic acid) (DTNB = Ellman’s reagent), and cellulose membrane dialysis tubing (MWCO ~14000) were obtained from Sigma-Aldrich. Polyethylene glycol dimaleimides (RAPP Polymere, PEG-(C2H4-mal)2, Mn = 2000 Da and 6000 Da) were used as crosslinking agents. Cell culture supplements were purchased from Biological Industries. CCK-8 assay (Cayman) was used in the cell proliferation studies.
Extraction and Reduction of the Keratin Proteins. Merino sheep wools were cleaned and defatted using previously reported methods.26,27 Soluble keratin proteins were obtained by treating 1 g cleaned and defatted wool with 50 mL 125 mM sodium sulfide solution at 40 ℃ by shaking at 150 rpm for 4 h. The keratin extraction solution was dialyzed against deionized water using pretreated dialysis membrane tubing. The dialysis medium was changed thrice a day, and the dialysis process was carried out for 3 days. Then, the keratin solution was lyophilized and stored at -20 ℃. Keratin proteins are exposed to the second reduction process to increase their free thiol content.28 500 mg lyophilized keratin was dissolved in 10 mL deionized water at pH 7.4 and room temperature. 200 mg DTT was added to the keratin solution to cleave the disulfide bonds, and the solution was stirred at room temperature overnight. Next, the reduced keratins were purified using a centrifugal filter (Amicon Ultra-15 with Ultracel membrane, 3 kDa MWCO). Keratin proteins were isolated by freeze-drying and stored at -20 ℃. DTNB assay was used to determine the free thiol groups of the keratin proteins.29 UV absorbance values at 412 nm were measured on a Shimadzu UV-2450 model UV-Vis spectrophotometer. Free thiol content was determined using the extinction coefficient of 14150 M-1cm-1 at 412 nm.30
Preparation and Characterization of the PEG-keratin Hydrogels. Chemically crosslinked PEG-keratin hydrogels were prepared by mixing the reduced keratin and PEG-(C2H4-mal)2 solutions prepared in degassed PBS (10 mM NaH2PO4 and 150 mM NaCl) at pH 7.4 or in degassed deionized water at neutral pH. Immediately formed viscous solutions were incubated at 37 ℃ overnight to promote gelation. In the hydrogel formulations, the concentration of functionalized PEGs was fixed as 5 w%. The keratin amount used was determined according to the thiol-to-maleimide molar ratio of ~1 : 0.58 ± 0.04.
Oscillatory rheology tests of the hydrogels were conducted by following the protocol given elsewhere.31 A Thermo Fisher Scientific HAAKE MARS model rheometer with a 3.5 cm diameter was used in the experiments.
The swelling behavior of the hydrogels was investigated by soaking the lyophilized hydrogels in excess PBS buffer at room temperature and comparing the dry weight (Wd) and the swollen weights (Ws) of the hydrogels measured at specified periods. The following equation was used to get the swelling ratio (SR) values:32

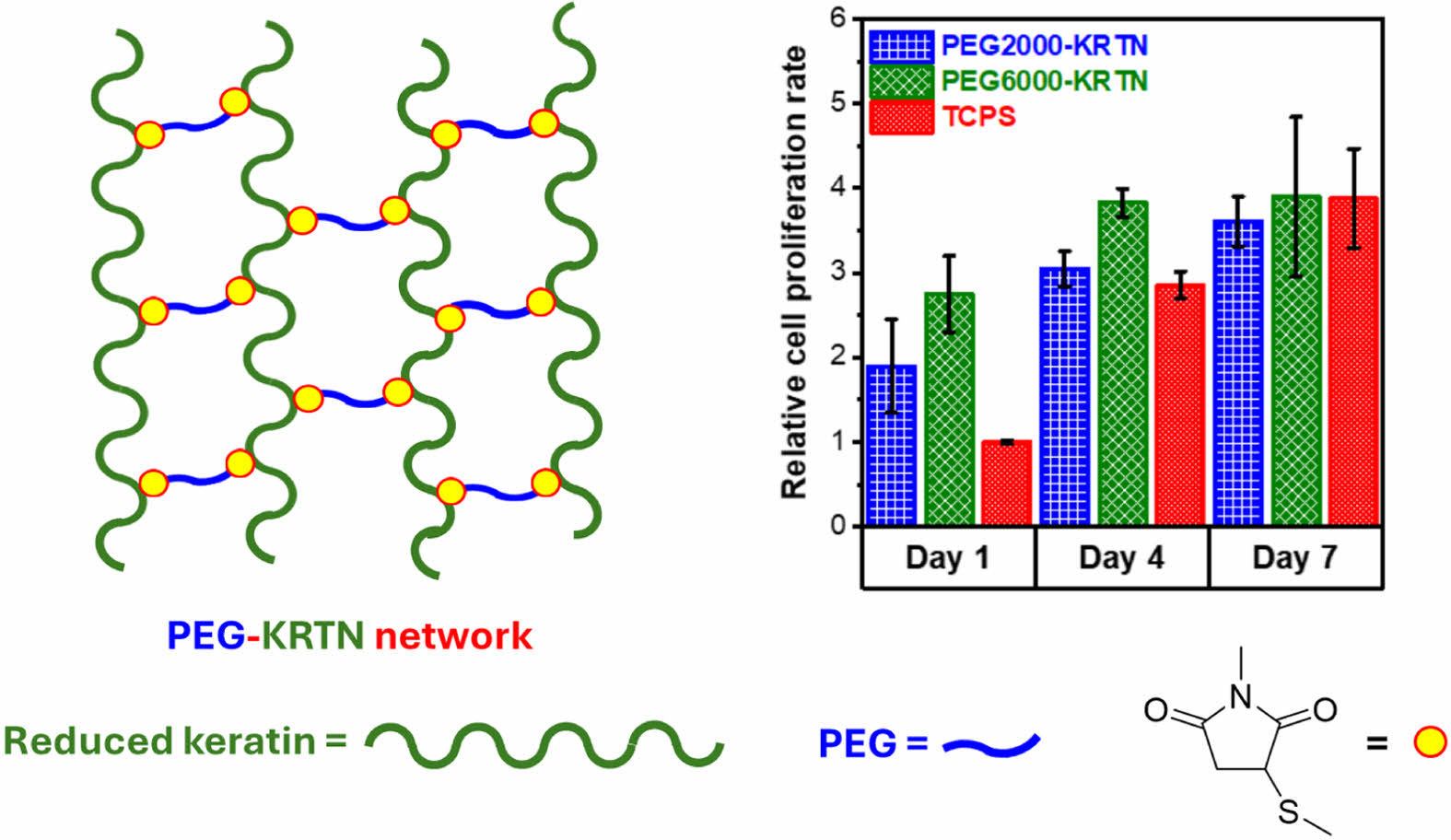
The pore structure of the hydrogels was determined using an FEI Quanta 250 FEG model scanning electron microscopy (SEM). The hydrogels prepared in deionized water were frozen in liquid nitrogen and freeze-dried at -80 ℃. A cross-sectionally fractured portion was observed after the sample was coated with gold. CCK-8 assay was used to investigate the cytocompatibility of the PEG-keratin hydrogels. CCK-8 analysis and cultivation of the L929 mouse fibroblasts were performed according to the previously used protocols.26,27 For the CCK-8 assay, absorbance measurements at 450 nm (A450) were taken with a Varioskan Flash model microplate reader. The relative cell proliferation rate (RCPR) values were determined using the following formula:27
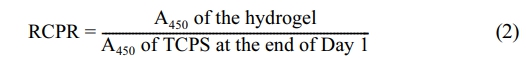
Keratin proteins were solubilized using sodium sulfide. The free thiol amount obtained in the sulfitolysis reaction is relatively low (~0.07 mmol/g keratin) due to the reformation of disulfide bonds during dialysis. For this reason, the keratin proteins were subjected to the second reduction process with DTT and purified very quickly using a centrifugal filter. Upon DTT reduction, the free thiol amount increased to 0.62 mmol/g keratin. Similar to our study, sodium sulfide was used to extract keratin proteins from human hair, and the free thiol amount of the keratin proteins was determined to be less than 0.01 mmol/g keratin. After the DTT reduction process, the free thiol content of the human hair keratins was reported as 0.55 mmol/g keratin.28
PEG-keratin hydrogels were prepared at a fixed functionalized PEG concentration of 5 w% and ~1 : 0.58 ± 0.04 thiol to maleimide ratio. At these conditions, the hydrogels with 12.5 w% and 10 w% total polymer concentration (keratin + functionalized PEG) were formulated for 2000 Da or 6000 Da PEG-(C2H4-mal)2 components and the resultant hydrogels were denoted as PEG2000-KRTN and PEG6000-KRTN, respectively. After mixing reduced keratin with 2000 Da or 6000 Da PEG-(C2H4-mal)2 at pH 7.4 and incubating at 37 ℃, self-standing hydrogels were obtained. As thiols were used in excess amounts, the network formation is mainly due to the Michael addition reaction and the formation of disulfide bonds upon oxidation (Scheme 1).
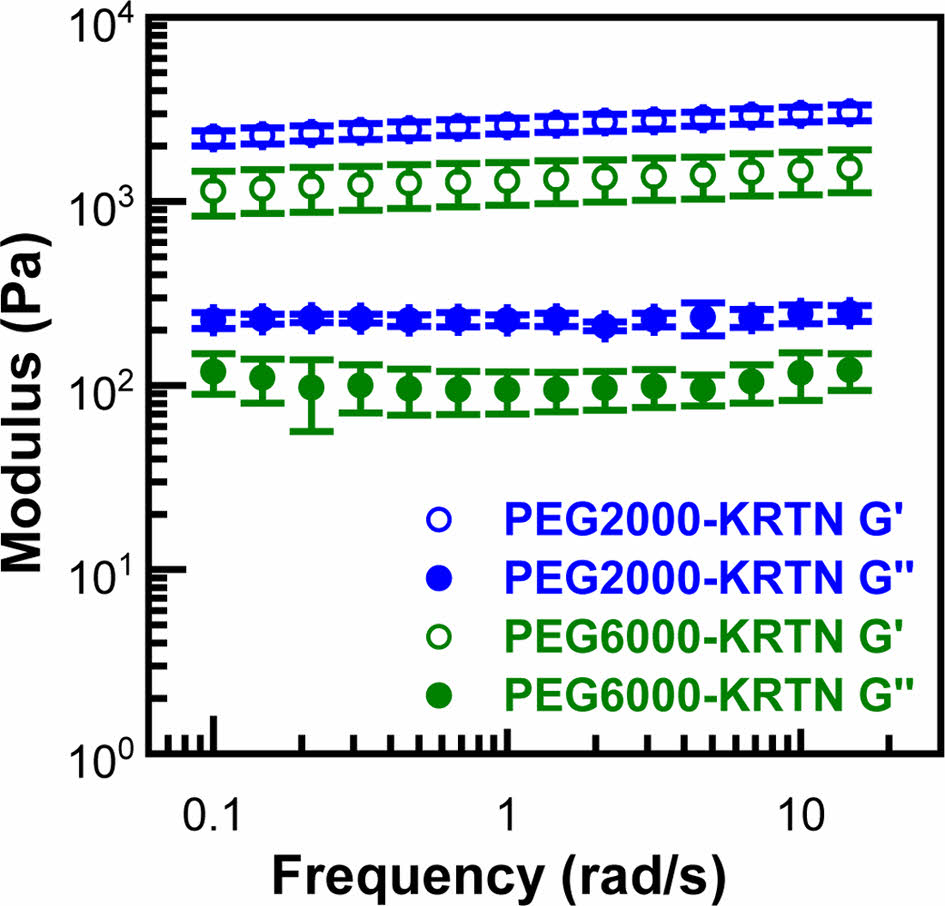
The viscoelastic behavior of the PEG-keratin hydrogels was investigated using frequency sweep, strain sweep, and cyclic strain time sweep experiments. First, the frequency sweep tests were done from 0.1 Hz to 15 Hz at a constant 0.2% strain, and the resultant curves are shown in Figure 1. Storage modulus (G') values of the PEG-keratin hydrogels were higher than those of the loss modulus (G'') values, confirming gel-like structures composed of the crosslinked viscoelastic network.33
Plateau values of storage moduli were determined as 2613 ± 254 and 1313 ± 345 Pa for PEG2000-KRTN and PEG6000-KRTN hydrogels, respectively. The higher stiffness of the PEG2000-KRTN hydrogel can be attributed to its higher number of crosslinking points, as the amount of keratin is higher in the PEG2000-KRTN hydrogel formulation. It is also possible that shorter and more mobile PEG2000-dimaleimide chains can react with the thiol groups more readily, contributing to the crosslinking density. G' values of the PEG-keratin hydrogels obtained in this study are much higher than those of the tetrakis (hydroxymethyl)
phosphonium chloride (THPC) crosslinked keratose hydrogels (between ~0.06 and 0.8 kPa) prepared at 7.5 w% biopolymer concentration.27
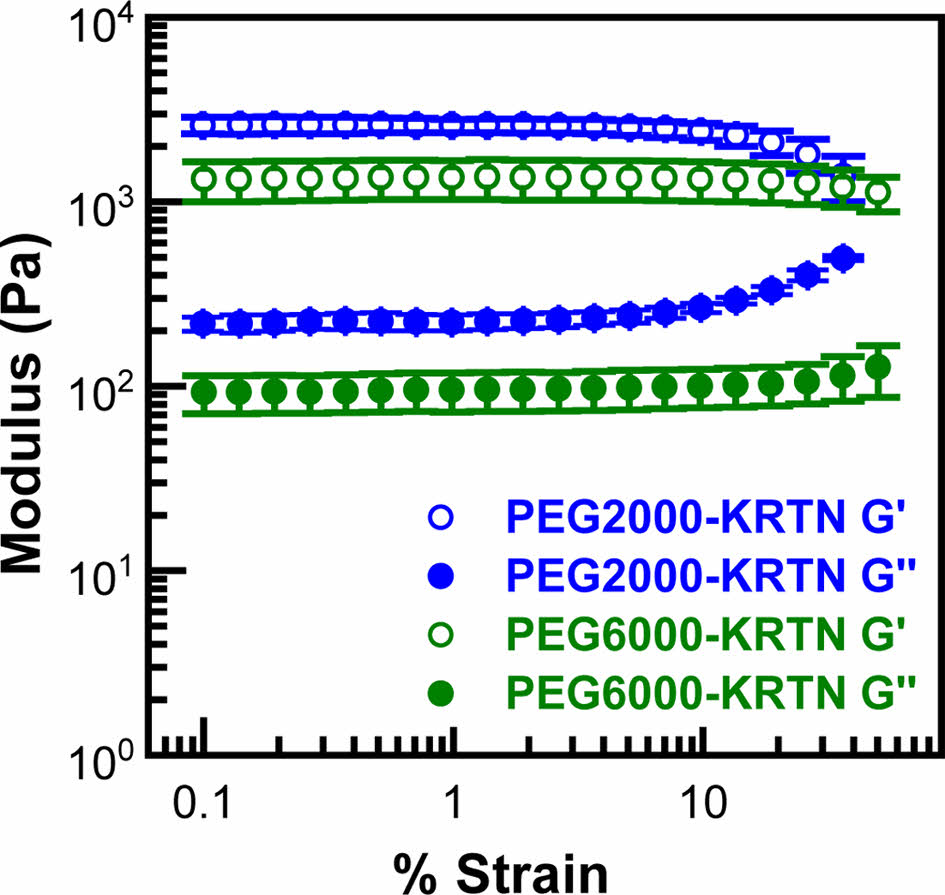
Linear viscoelastic regions (LVER) of the hydrogels were determined using strain sweep data given in Figure 2. LVER of the PEG2000-KRTN hydrogel was observed up to 10% strain. However, PEG6000-KRTN hydrogel exhibited a broader range of LVER with a 40% critical strain value. After these critical strain values, G' values decreased, and G'' values started to increase, indicating an increase in the viscous behavior and destruction of the network structure of the hydrogels.34 The higher critical strain of the PEG6000-KRTN hydrogel can be attributed to its lower crosslinking density.35
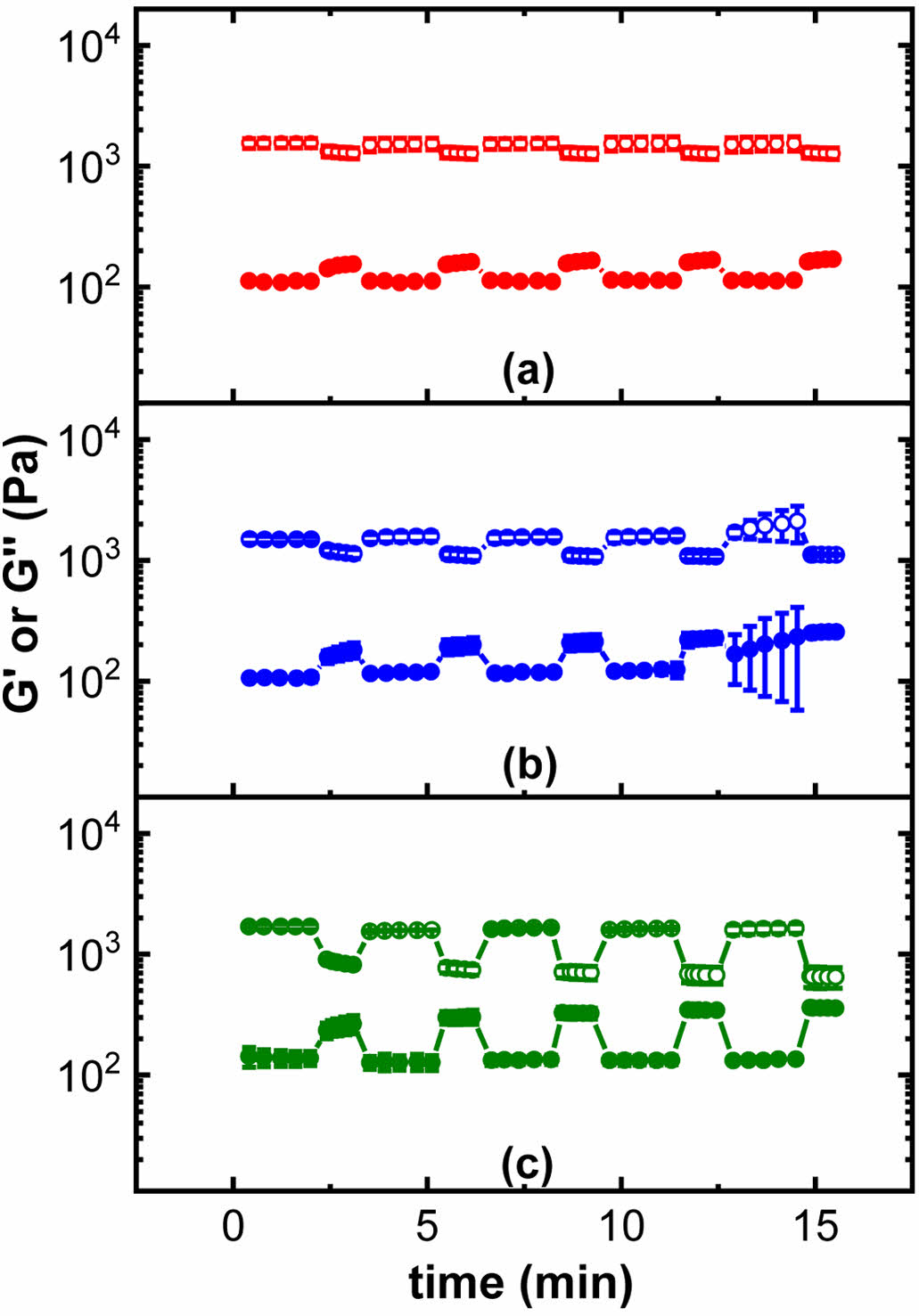
Cyclic time sweep experiments were performed using 40%, 60% and 100% strains outside the LVER region of the hydrogels. The PEG2000-KRTN hydrogel structure failed upon exposing high strain values. PEG6000-KRTN hydrogel, on the other hand, restored its structure and did not show elastic to viscous transition up to 100% strain, confirming its more elastic nature (Figure 3).
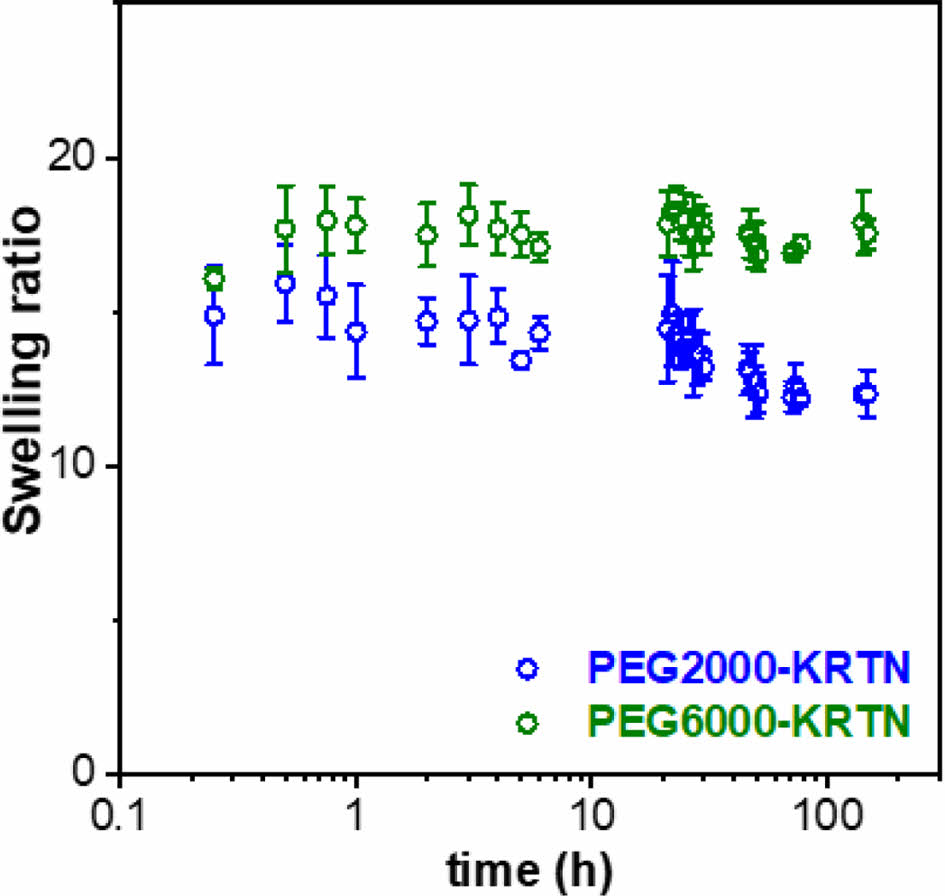
The swelling kinetic curves of the PEG-hydrogels are shown in Figure 4. The average swelling ratios of the PEG2000-KRTN and PEG6000-KRTN hydrogels were determined as 13.7 ± 1.0 and 17.6 ± 0.5, respectively. The lower swelling ratio of PEG2000-KRTN hydrogel can be explained by its stiffness, which restricts water penetration into the network structure.36,37
Both hydrogels maintained their structures for up to 6 days. After six days, the samples started to dissolve in the PBS buffer.
Figure 5 shows SEM images of freeze-dried hydrogels. A highly irregular pore structure with open and closed pores was obtained for both hydrogels. PEG6000-KRTN hydrogel exhibited a more porous structure, which can be attributed to the higher molar mass of PEG and its lower number of crosslinking points. Additionally, fibrillar structures around the pores and pore walls of the PEG6000-KRTN hydrogel were observed.
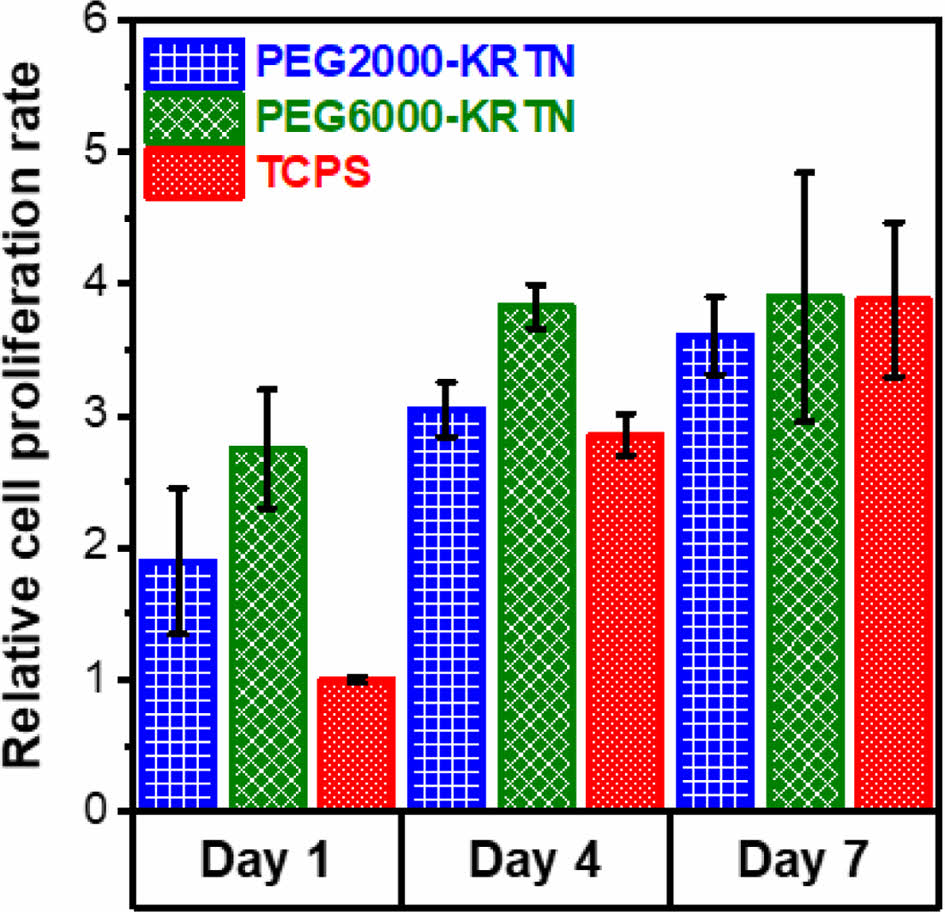
Cell proliferation tests were performed to examine the interactions between the hydrogels and the L929 mouse fibroblast cell line using CCK-8 assay. Relative cell proliferation rate results are given in Figure 6. For day 1 and day 4, both hydrogels promoted cell proliferation at a rate higher than tissue culture polystyrene (TCPS). However, THPC crosslinked keratin hydrogels prepared by our group exhibited lower cell proliferation than TCPS.27
The superior biocompatibility of the PEG-keratin hydrogels can be attributed to their higher stiffness and larger pore sizes. Between the fourth and seventh days, slower cell proliferation was obtained, suggesting the saturation of the seeded surfaces by the cells.
|
Figure 1 Frequency sweep plots of the hydrogels. |
|
Figure 2 Strain sweep plots of the hydrogels. |
|
Figure 3 Cyclic strain time sweep plots of the PEG6000-KRTN hydrogel exposed to 0.2% low strain followed by (a) 40%; (b) 60%; (c) 100% high strain. G' = open circles, G'' = closed circles. |
|
Figure 4 Swelling behaviors of the freeze-dried PEG-KRTN hydrogels. |

|
Figure 5 SEM images of the freeze-dried: (a) PEG2000-KRTN; (b) PEG6000-KRTN hydrogels. Scale bars: 500 μm. |
|
Figure 6 Cell proliferation results of the PEG-KRTN hydrogels. |
PEG-keratin hydrogels were successfully prepared by applying simple thiol-maleimide chemistry. The initial solubilization of keratin proteins using sodium sulfide generated a relatively small number of thiols, but the subsequent reduction with DTT significantly increased the thiol content of the keratin. Using different molar masses of PEG dimaleimides (2000 Da and 6000 Da) in the crosslinking reaction profoundly affected the mechanical and physicochemical properties of the resultanthydrogels. The PEG2000-KRTN hydrogels presented higher stiffness due to increased crosslinking points, whereas the PEG6000-KRTN hydrogels exhibited higher elasticity, attributed to the longer PEG chains and lower crosslinking density. Swelling experiments showed that the PEG6000-KRTN hydrogel had a higher swelling ratio, correlating with its more porous structure, as revealed by its SEM image. PEG-keratin hydrogels promoted a higher cell proliferation rate than the TCPS surfaces, confirming their biocompatibility. The results indicate that these hydrogels with tunable properties can find applications in soft tissue engineering.
- 1. Stowers, R. S. Advances in Extracellular Matrix-mimetic Hydrogels to Guide Stem Cell Fate. Cells Tissues Organs 2022, 211, 703-720.
-

- 2. Murphy, N. P.; Lampe, K. J. Mimicking Biological Phenomena in Hydrogel-based Biomaterials to Promote Dynamic Cellular Responses. J. Mater. Chem. B 2015, 3, 7867-7880.
-

- 3. Yang, F.; Williams, C. G.; Wang, D.; Lee, H.; Manson, P. N.; Elisseeff, J. The Effect of Incorporating RGD Adhesive Peptide in Polyethylene Glycol Diacrylate Hydrogel on Osteogenesis of Bone Marrow Stromal Cells. Biomaterials 2005, 26, 5991-5998.
-

- 4. González-Pérez, F.; Alonso, M.; Torre, I.; Santos, M.; Rodríguez-Cabello, J. Laminin-Derived Peptide Sequences Within Elastin-Like Recombinamer Scaffolds Provide Spatiotemporally Synchronized Guidance of Angiogenesis and Neurogenesis. Adv. Healthc. Mater 2022, 11, 2201646.
-

- 5. Perera, T. H.; Lu, X.; Smith Callahan, L. A. Effect of Laminin Derived Peptides IKVAV and LRE Tethered to Hyaluronic Acid on HiPSC Derived Neural Stem Cell Morphology, Attachment and Neurite Extension. J. Funct. Biomater. 2020, 11, 15.
-

- 6. Wei, X.; Chen, S.; Xie, T.; Chen, H.; Jin, X.; Yang, J.; Sahar, S.; Huang, H.; Zhu, S.; Liu, N. An MMP-degradable and Conductive Hydrogel to Stabilize HIF-1α for Recovering Cardiac Functions. Theranostics 2022, 12, 127.
-

- 7. Charrier, E. E.; Pogoda, K.; Wells, R. G.; Janmey, P. A. Control of Cell Morphology and Differentiation by Substrates with Independently Tunable Elasticity and Viscous Dissipation. Nat. Commun. 2018, 9, 449.
-

- 8. Chester, D.; Lee, V.; Wagner, P.; Nordberg, M.; Fisher, M. B.; Brown, A. C. Elucidating the Combinatorial Effect of Substrate Stiffness and Surface Viscoelasticity on Cellular Phenotype. J. Biomed. Mater. Res. A 2022, 110, 1224-1237.
-

- 9. Fan, Y.; Sun, Q.; Li, X.; Feng, J.; Ao, Z.; Li, X.; Wang, J. Substrate Stiffness Modulates the Growth, Phenotype, and Chemoresistance of Ovarian Cancer Cells. Front. Cell Dev. Biol. 2021, 9, 718834.
-

- 10. Lu, C.; Zheng, J.; Yoshitomi, T.; Kawazoe, N.; Yang, Y.; Chen, G. How Hydrogel Stiffness Affects Adipogenic Differentiation of Mesenchymal Stem Cells under Controlled Morphology. ACS Appl. Bio Mater. 2023, 6, 3441-3450.
-

- 11. Nguyen, D. H.; Utama, R. H.; Tjandra, K. C.; Suwannakot, P.; Du, E. Y.; Kavallaris, M.; Tilley, R. D.; Gooding, J. J. Tuning the Mechanical Properties of Multiarm RAFT-Based Block Copolyelectrolyte Hydrogels via Ionic Cross-Linking for 3D Cell Cultures. Biomacromolecules 2022, 24, 57-68.
-

- 12. Guaresti, O.; Basasoro, S.; González, K.; Eceiza, A.; Gabilondo, N. In situ Cross–linked Chitosan Hydrogels via Michael Addition Reaction Based on Water–soluble Thiol–maleimide Precursors. Eur. Polym. J. 2019, 119, 376-384.
-

- 13. Moon, N. G.; Pekkanen, A. M.; Long, T. E.; Showalter, T. N.; Libby, B. Thiol-Michael ‘click’ Hydrogels as an Imageable Packing Material for Cancer Therapy. Polymer 2017, 125, 66-75.
-

- 14. Hebner, T. S.; Kirkpatrick, B. E.; Fairbanks, B. D.; Bowman, C. N.; Anseth, K. S.; Benoit, D. S. Radical-Mediated Degradation of Thiol–Maleimide Hydrogels. Adv. Sci. 2024, 2402191.
-

- 15. Jansen, L. E.; Negrón-Piñeiro, L. J.; Galarza, S.; Peyton, S. R. Control of Thiol-maleimide Reaction Kinetics in PEG Hydrogel Networks. Acta Biomater. 2018, 70, 120-128.
-

- 16. Altinbasak, I.; Kocak, S.; Sanyal, R.; Sanyal, A. Fast-Forming Dissolvable Redox-Responsive Hydrogels: Exploiting the Orthogonality of Thiol–Maleimide and Thiol–Disulfide Exchange Chemistry. Biomacromolecules 2022, 23, 3525-3534.
-

- 17. Kharkar, P. M.; Kiick, K. L.; Kloxin, A. M. Design of Thiol-and Light-sensitive Degradable Hydrogels Using Michael-type Addition Reactions. Polym. Chem. 2015, 6, 5565-5574.
-

- 18. Pupkaite, J.; Rosenquist, J.; Hilborn, J.; Samanta, A. Injectable Shape-holding Collagen Hydrogel for Cell Encapsulation and Delivery Cross-linked Using Thiol-michael Addition Click Reaction. Biomacromolecules 2019, 20, 3475-3484.
-

- 19. García, A. J. PEG–maleimide Hydrogels for Protein and Cell Delivery in Regenerative Medicine. Ann. Biomed. Eng. 2014, 42, 312-322.
-

- 20. Sarma, A. Biological Importance and Pharmaceutical Significance of Keratin: A Review. Int. J. Biol. Macromol. 2022, 219, 395-413.
-

- 21. Lazarus, B. S.; Chadha, C.; Velasco-Hogan, A.; Barbosa, J. D.; Jasiuk, I.; Meyers, M. A. Engineering with Keratin: A Functional Material and a Source of Bioinspiration. iScience 2021, 24.
-

- 22. Wang, X.; Shi, Z.; Zhao, Q.; Yun, Y. Study on the Structure and Properties of Biofunctional Keratin from Rabbit Hair. Materials 2021, 14, 379.
-

- 23. Giteru, S. G.; Ramsey, D. H.; Hou, Y.; Cong, L.; Mohan, A.; Bekhit, A. E. D. A. Wool Keratin as a Novel Alternative Protein: A Comprehensive Review of Extraction, Purification, Nutrition, Safety, and Food Applications. Compr. Rev. Food Sci. Food Saf. 2023, 22, 643-687.
-

- 24. Silva, R.; Singh, R.; Sarker, B.; Papageorgiou, D. G.; Juhasz, J. A.; Roether, J. A.; Cicha, I.; Kaschta, J.; Schubert, D. W.; Chrissafis, K. Hybrid Hydrogels Based on Keratin and Alginate for Tissue Engineering. J. Mater. Chem. B 2014, 2, 5441-5451.
-

- 25. Tachibana, A.; Furuta, Y.; Takeshima, H.; Tanabe, T.; Yamauchi, K. Fabrication of Wool Keratin Sponge Scaffolds for Long-term Cell Cultivation. J. Biotechnol. 2002, 93, 165-170.
-

- 26. Pakkaner, E.; Yalçın, D.; Uysal, B.; Top, A. Self-assembly Behavior of the Keratose Proteins Extracted from Oxidized Ovis Aries Wool Fibers. Int. J. Biol. Macromol. 2019, 125, 1008-1015.
-

- 27. Yalçın, D.; Top, A. Novel Biopolymer-based Hydrogels Obtained Through Crosslinking of Keratose Proteins Using Tetrakis (hydroxymethyl) Phosphonium Chloride. Iran. Polym. J. 2022, 31, 1057-1067.
-

- 28. Yue, K.; Liu, Y.; Byambaa, B.; Singh, V.; Liu, W.; Li, X.; Sun, Y.; Zhang, Y. S.; Tamayol, A.; Zhang, P. Visible Light Crosslinkable Human Hair Keratin Hydrogels. Bioeng. Transl. Med. 2018, 3, 37-48.
-

- 29. Riddles, P. W.; Blakeley, R. L.; Zerner, B. Reassessment of Ellman's Reagent. In Methods in Enzymology, C. H. W. Hirs; Timasheff, S. N., Eds. Academic Press: New York, 1983, 91, 49-60.
-

- 30. Aitken, A.; Learmonth, M. Estimation of Disulfide Bonds Using Ellman’s Reagent. In The Protein Protocols Handbook, Walker, J. M., Ed. Humana Press: Totowa, NJ, 2009; 1053-1055.
-

- 31. Chen, M. H.; Wang, L. L.; Chung, J. J.; Kim, Y. H.; Atluri, P.; Burdick, J. A. Methods to Assess Shear-thinning Hydrogels for Application as Injectable Biomaterials. ACS Biomater. Sci. Eng. 2017, 3, 3146-3160.
-

- 32. Kim, B.; Kim, J. PEGylated Hybrid Hydrogels with Tunable Swelling Property and Biodegradability. Polym. Korea 2021, 45, 171-177.
-

- 33. Ozbas, B.; Kretsinger, J.; Rajagopal, K.; Schneider, J. P.; Pochan, D. J. Salt-triggered Peptide Folding and Consequent Self-assembly Into Hydrogels with Tunable Modulus. Macromolecules 2004, 37, 7331-7337.
-

- 34. Karvinen, J.; Ihalainen, T. O.; Calejo, M. T.; Jönkkäri, I.; Kellomäki, M. Characterization of the Microstructure of Hydrazone Crosslinked Polysaccharide-based Hydrogels Through Rheological and Diffusion Studies. Mater. Sci. Eng. C 2019, 94, 1056-1066.
-

- 35. Gu, S.; Cheng, G.; Yang, T.; Ren, X.; Gao, G. Mechanical and Rheological Behavior of Hybrid Cross-linked Polyacrylamide/Cationic Micelle Hydrogels. Macromol. Mater. Eng. 2017, 302, 1700402.
-

- 36. Feng, W.; Wang, Z. Tailoring the Swelling-Shrinkable Behavior of Hydrogels for Biomedical Applications. Adv. Sci. 2023, 10, 2303326.
-

- 37. Hoti, G.; Caldera, F.; Cecone, C.; Rubin Pedrazzo, A.; Anceschi, A.; Appleton, S. L.; Khazaei Monfared, Y.; Trotta, F. Effect of the Cross-linking Density on the Swelling and Rheological Behavior of Ester-bridged β-cyclodextrin Nanosponges. Materials 2021, 14, 478.
-

- Polymer(Korea) 폴리머
- Frequency : Bimonthly(odd)
ISSN 2234-8077(Online)
Abbr. Polym. Korea - 2024 Impact Factor : 0.6
- Indexed in SCIE
 This Article
This Article
-
2025; 49(2): 163-169
Published online Mar 25, 2025
- 10.7317/pk.2025.49.2.163
- Received on Jun 19, 2024
- Revised on Sep 30, 2024
- Accepted on Nov 10, 2024
 Services
Services
- Full Text PDF
- Abstract
- ToC
- Acknowledgements
- Conflict of Interest
Introduction
Experimental
Results and Discussion
Conclusions
- References
Shared
 Correspondence to
Correspondence to
- Ayben Top
-
Department of Chemical Engineering, İzmir Institute of Technology, Urla, İzmir, 35430, Türkiye
- E-mail: aybentop@iyte.edu.tr
- ORCID:
0000-0003-0554-2506









 Copyright(c) The Polymer Society of Korea. All right reserved.
Copyright(c) The Polymer Society of Korea. All right reserved.