- Gas-phase Copolymerization of Ethylene, Propene and Hydrogen under Controlled Atmosphere Composition in Reactor
Zhejiang Industrial Polytechnic College, 151 Qutun Road, Yuecheng District, Shaoxing, Zhejiang 312000, China
- 반응기의 제어된 대기 조성 하에서 에틸렌, 프로펜 및 수소의 가스상 공중합
Reproduction, stored in a retrieval system, or transmitted in any form of any part of this publication is permitted only by written permission from the Polymer Society of Korea.
The successful application of olefin gas-phase polymerization processes shows the progress of polymerization processes and reactor design. Detection and control of the atmosphere composition in the gas phase is of great importance to design of gas-phase polymerization reactor, especially for copolymerization reactor, because the atmosphere composition in the gas phase has an important influence on the polymerization kinetics and the properties of the polymers obtained. In this work, a method for controlling the atmosphere composition during gas-phase copolymerization of ethylene, propene and hydrogen was developed. Ethylene/propene gas-phase copolymerization with little hydrogen during synthesis of PP/P(E-co-P) in-reactor alloy was carried out, and the atmosphere composition was controlled at an almost constant level. The effect of the atmosphere composition in the reactor on gas-phase polymerization was investigated and the reactivity ratios of ethylene and propene were estimated.
A method for controlling the atmosphere composition during gas-phase copolymerization of ethylene, propene, and hydrogen was developed. The atmosphere composition was maintained constant, and the effects on polymerization behavior and polymer properties were investigated. The reactivity ratios of ethylene and propene were estimated, providing insights into the copolymerization kinetics.
Keywords: olefin copolymerization, gas-phase polymerization, atmosphere composition control, reactivity ratios.
The research was supported by Key Investment Plan for High-level Talents of Zhejiang Industrial Polytechnic College under award No. 112709010921621119 and Zhejiang Provincial Natural Science Foundation under award No. LGG20E030001.
The authors declare that there is no conflict of interest.
Supporting information gives (a) atmosphere compositions in the reactor and monomer consumption rates during gas-phase copolymerization in runs 7 and 2-4, (b) GPC curves, 13C NMR spectra and DSC heating curves of the polymers in runs 1-6 and (c) GPC, DSC, SSA and 13C NMR analysis of Fractions A-C of the polymer samples in runs 1-4. The materials are available via the Internet at https://journal.polymer-korea.or.kr.
PK_2025_049_02_187_Supporting_Information.pdf (1,250 kb)
Supplementary Information
Olefin gas-phase polymerization processes, such as UnipolTM, Spherilene,1 Borstar® PE,2 Innovene,3 Catalloy,4 and Spherizone,5 have been successfully applied to the commercial production of polyethylenes and polypropylenes since the end of 1960s. Several advantages of gas-phase processes6 are: (a) elimination of the costs associated with storage and treatment of large quantities of solvents, (b) simplification of the polymerization systems, (c) reduction of environmental problems due to lack of solvents, (d) ease of preparing ethylene/propylene block copolymers, (e) no monomer or hydrogen solubility problems and (f) no additional cost to dry polymers. The successful application of gas-phase processes proves the progress made in polymerization processes and reactor design.7
For design of gas-phase polymerization reactor, it is necessary to determine and control the atmosphere composition (monomer concentrations) in the reactor,8 especially when copolymerization is involved. This is because the atmosphere composition in the reactor has an important influence on the polymerization kinetics and the properties of the polymers obtained.9-12 The papers in the open literature on gas-phase polymerization reactors capable of detecting or controlling atmosphere composition were mainly based on two different methods, one was gas chromatography (GC),8,13-17 the other was Fourier transform infrared spectroscopy (FTIR).18-21 The reactor systems constructed allowed the analysis or control of the atmosphere composition in the reactor, which was very important for the study of the influence of the atmospheric composition on the properties of polymers. At the same time, the
monomer consumption rates were also collected, which were helpful to the study on polymerization kinetics.
The monomer reactivity ratios are important kinetic parameters in Mayo-Lewis copolymer composition equation.22 They can be used to evaluate the copolymerization ability of the catalyst and determine the composition and sequence distribution of the copolymers.23,24 Han-Adebekun25 and Tan15 estimated the monomer reactivity ratios in ethylene/propene gas-phase copolymerization, when the controlled atmosphere composition in the reactor was constant. According to Fineman- Ross equation, the reactivity ratios were evaluated by the copolymer composition and feed composition, in which the copolymer composition was determined by the integrals of monomer consumption rates25 or 13C NMR.15
In our previous work,26 based on the series connection of a Coriolis flowmeter, a thermal flowmeter and a differential pressure flowmeter, a new method for detecting the atmosphere composition in the process of ethylene, propylene and hydrogen gas phase copolymerization was proposed, where hydrogen plays a pivotal role in the copolymerization process, as it regulates molecular weight, influences catalyst activity, and controls the morphology of in-reactor PP/P(E-co-P) alloys, making it an indispensable component in the industrial production process. With this method, ethylene/propene gas-phase copolymerization with hydrogen modulation was carried out in the process of preparing PP/P(E-co-P) in-reactor alloy. The atmosphere composition in the reactor was detected accurately and sensitively, and the monomer consumption rates were deduced according to the mass balance.
In this paper, the method for controlling the atmosphere composition during gas-phase copolymerization of ethylene, propene and hydrogen was further developed. In the process of synthesizing PP/P(E-co-P) in-reactor alloy, ethylene/propene gas-phase copolymerization with hydrogen modulation was carried out when the atmosphere composition in the reactor was controlled constant. The effects of atmosphere composition on polymerization behavior and polymer properties were studied, and the reactivity ratios of ethylene and propene were estimated.
The experimental materials and procedure were similar to those in our previous article.26 For the convenience of the reader, it is repeated here.
Materials and Devices. Hydrogen (Hangzhou Jingong Special Gas Co., Ltd., 99.99%), Ethylene (Sinopec Beijing Research Institute of Chemical Industry, Polymerization-grade) and Propene (Hangzhou Minxing Gas Co., Ltd., 99.99%) passed through a gas purification box to remove trace water and oxygen and then were injected into the reactor. The catalyst, TiCl4/MgCl2/9,9-bis(methoxymethyl) fluorene with a titanium content of 5.76 wt%, supplied by Renqiu LIHE Technology Ltd., serves as a vital heterogeneous catalyst in gas-phase polymerization. It facilitates the adsorption and polymerization of olefin monomers, ultimately yielding polymers with tailored properties. This catalyst is indispensable for achieving the desired structural, morphological, and performance characteristics through the optimization of reaction conditions, such as pressure and monomer concentrations. Methyl cyclohexyl dimethoxysilane (C-Donor, Renqiu LIHE Technology Ltd., 99.0%) was utilized as 0.1 mol L-1 toluene solution and aluminumtriethyl (TEA, Albemarle, 93.0%) as 1 mol L-1 toluene solution. Toluene (Sinopharm Chemical Reagent Co., Ltd., AR) and n-Hexane (Sinopharm Chemical Reagent Co., Ltd., AR) passed through the solvent purification apparatus (MBRAUN, MB-SPS) and then were stored with 4 Å molecular sieves under nitrogen. All moisture- or air-sensitive compounds were treated in a glovebox under high purity nitrogen.
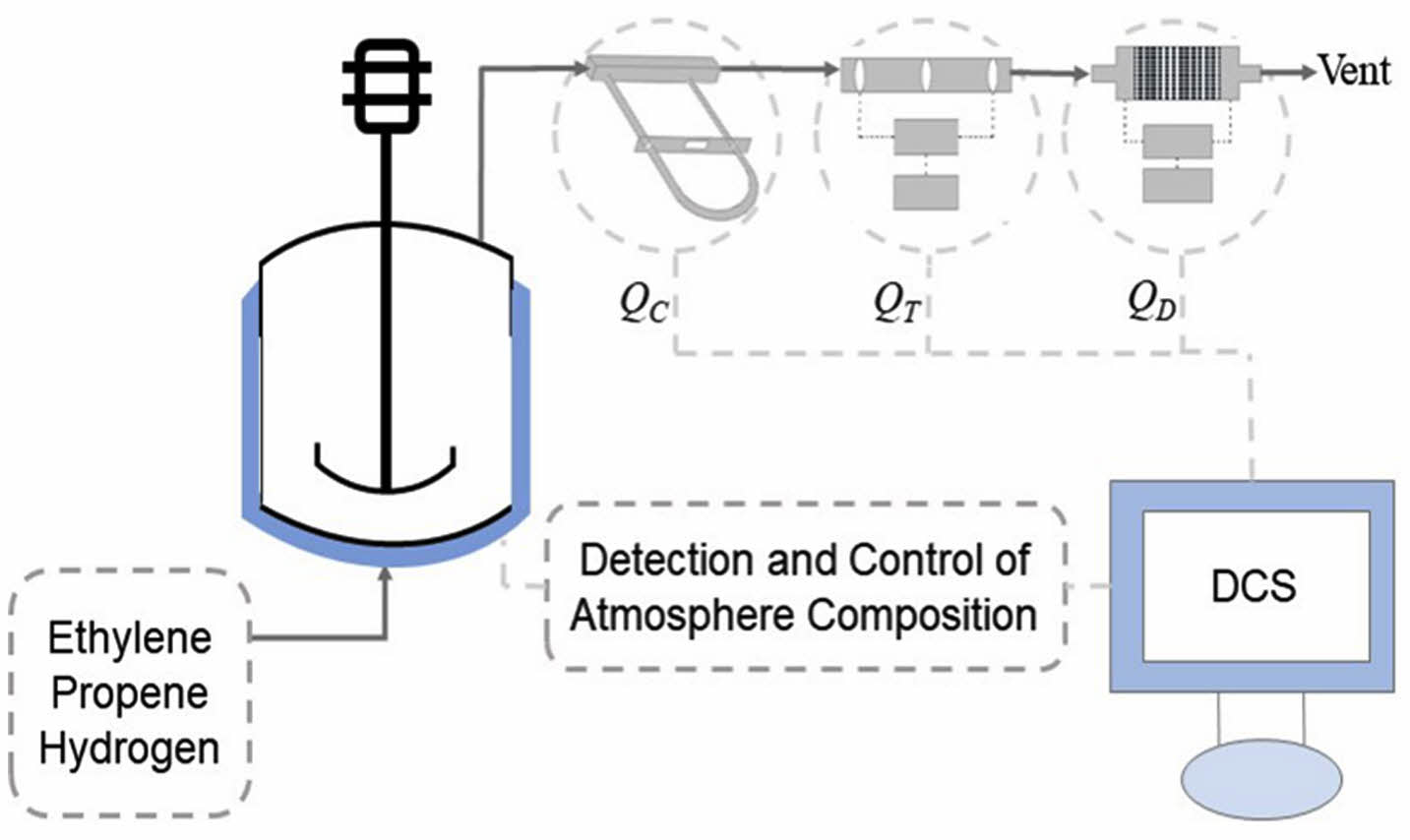
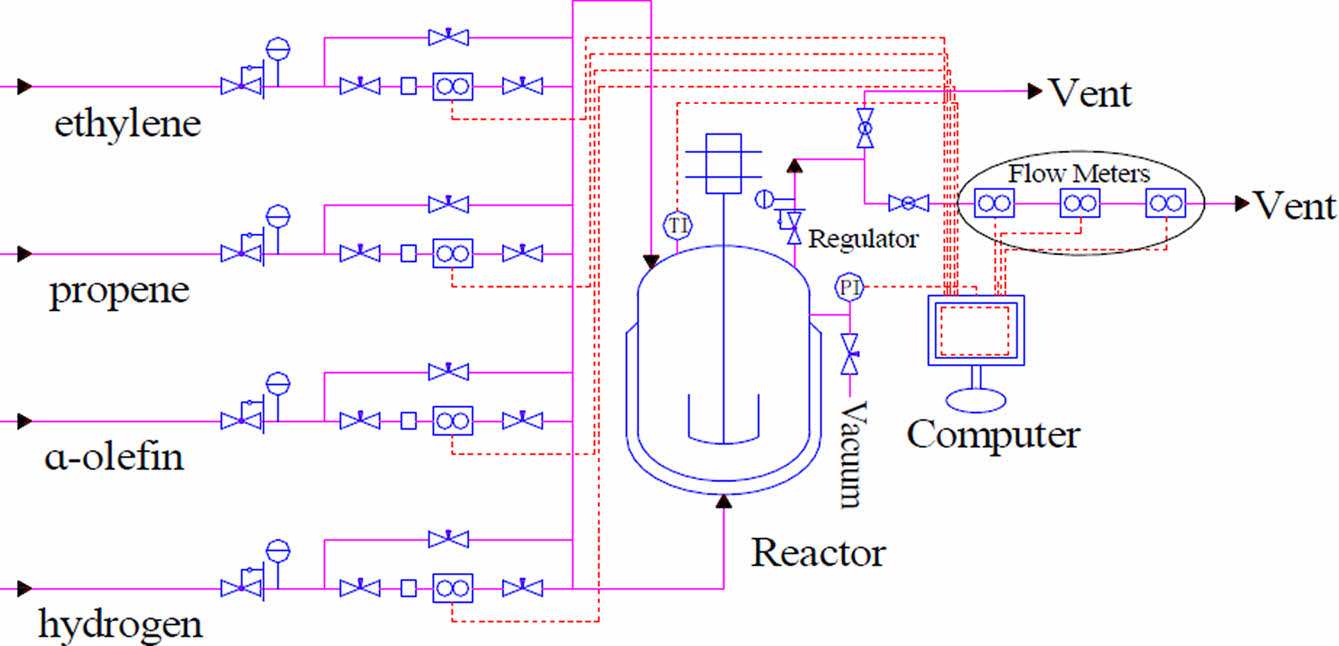
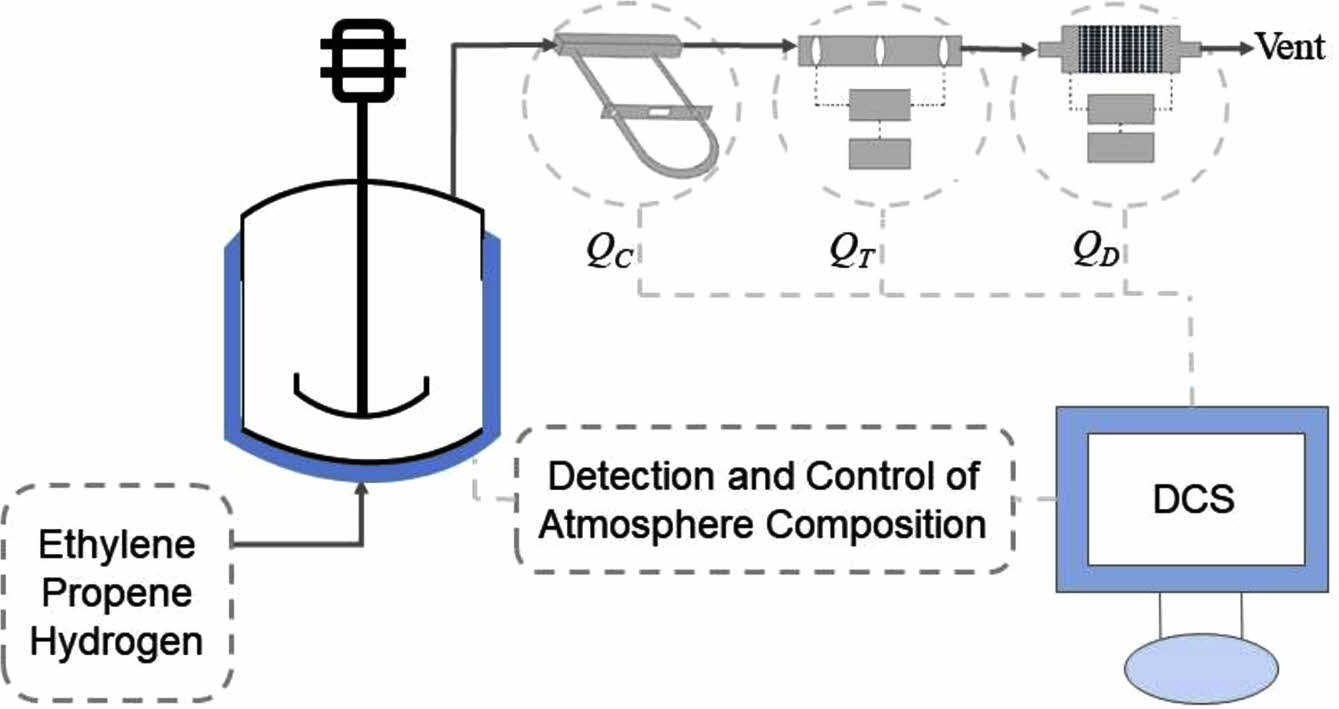
As shown in Figure 1, gas intake lines are equipped with Swagelok reduction valves and thermal mass flow controllers (Brooks Instruments, SLA5850). Ethylene and propene flow controllers are calibrated with nitrogen and the standard pressure and temperature are selected at 1 atm and 20 ℃. Hydrogen flow controller is calibrated with hydrogen and the standard pressure and temperature are selected at 1 atm and 0 ℃. The three lines converge at one point and are connected to the top and bottom of the reactor. The top of the reactor is connected with a vent line, which is connected in series with a Swagelok reduction valve, a Coriolis mass flowmeter (Brooks Instruments, Quantim), a thermal mass flowmeter (Brooks Instruments, SLA5800) and a differential pressure flowmeter (Alicat Scientific, Alicat MC). The Coriolis flowmeter is calibrated with air. The thermal flowmeter on the vent line is calibrated with ethylene and the standard pressure and temperature are selected at 1 atm and 0 ℃. The differential pressure flowmeter is calibrated with air and the standard pressure and temperature are selected at 1 atm and 25 ℃. The measurement accuracy of the Coriolis flowmeter, thermal flowmeter and differential pressure flowmeter is ±0.5%, ±0.18% and ±0.4%, respectively. The response time of the Coriolis flowmeter, thermal flowmeter and differential pressure flowmeter is below 0.5 s, 1 s and 0.1 s, respectively. The reactor temperature, pressure, gas feed flow and real-time volume flow readings of the three flow meters on the vent line can be monitored via a computer equipped with a distributed control system (DCS, SUPCON). The temperature and pressure of the exhaust are also monitored because temperature and pressure sensors are embedded in the differential pressure flowmeter.
The DCS can control the flow controllers on the intake lines to inject gases into the reactor in two modes. The first mode (Mode I) was that gases were controlled to flow into the reactor at a constant volume flow ratio, which was reported in our previous article.26 The second mode (Mode II) was that gases were injected into the reactor to maintain a constant atmosphere composition (monomer molar ratio), which is on the basis of the atmosphere composition detection process in the previous work.26 Figure 2 shows the atmosphere composition control process. When gas mixture of ethylene, propene and hydrogen entered the Coriolis flowmeter, the thermal flowmeter and the differential pressure flowmeter on the vent line from the top of the reactor, the three flowmeters output different volume flow readings. The atmosphere composition could be calculated by the computer according to a set of equations reported in our previous article:26
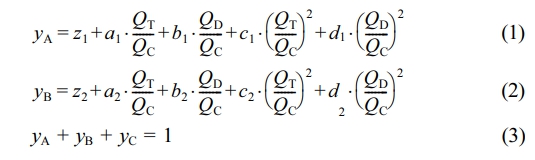
where QC, QT and QD are the volume flow readings of the Coriolis flowmeter, thermal flowmeter and differential pressure flowmeter, yA, yBand yCare the molar fractions of ethylene, propene and hydrogen. And z1= 3.20977, a1= -4.26232, b1= 4.17147, c1= 0.58209, d1= -1.26432, z2= -2.46663, a2= 5.40595, b2= -4.56239, c2= -1.23629, d2= 1.54756. By utilizing the real-time atmosphere composition of the vented exhaust, DCS feedback controlled the flow controllers on the intake lines to feed each gas to ensure that the atmosphere composition in the reactor reached the set value.
Polymerization Procedure. Ethylene and propene gas-phase copolymerization were performed in the presence of polypropylene particles produced in the first stage. Before use, the reactor was heated to 90 ℃, emptied and replaced with propene at least six times to ensure that the reactor was completely dry. In the first stage, the reactor was cooled down to 40 ℃ under propene atmosphere, and then n-hexane, TEA and C-Donor with specified volume were added. The catalyst was loaded into the reactor to initiate the polymerization, and immediately propylene was continuously supplied (through lines connected to the top of the reactor) to maintain a constant reactor pressure. After the preset time, the propene feed was closed and the solvent was emptied by evacuation. The mixture of ethylene, propene and hydrogen (through lines connected to the bottom of the reactor) were introduced to initiate the gas-phase polymerization. When the reactor pressure rose to a set value (about 8 minutes later), the three flowmeters on the vent line were opened manually, and the valve openings were 50%, 100% and 100%, respectively. After the preset time, the gas inlet valves were closed to stop the polymerization, the reactor was vented to atmospheric pressure, and the polymer was poured into acidified alcohol (hydrochloric acid content of 2 vol%). The suspension was stirred overnight, filtered and dried at 60 ℃ for 8 hours in vacuum.
Polymer Characterization. Molecular weight and molecular weight distribution of the polymers were determined by gel permeation chromatography (GPC) at 150 ℃ using a PL-GPC 220 system, which was coupled with an in-line capillary viscometer. The column was calibrated using the monodisperse polystyrene (PS) standards. The Mark-Houwink constants used for PS were K = 1.21 × 10-4 and α = 0.707, and for polypropene were K = 1.56 × 10-4 and α = 0.76. 1,2,4-Trichlorobenzene was used as solvent with a flow rate of 1.0 mL min-1.
The copolymer composition was measured by 13C NMR spectra at 120 ℃ with a Varian mercury 300-plus spectrometer. 10 wt% deuterated o-dichlorobenzene solutions of polymers were prepared by stirring for 3-4 hours at 150 ℃. Instrument conditions of quantitative NMR were optimized, including 90° pulse angle, inverse gated proton decoupling, 3 s pulse delay time and 0.8 s acquisition time. About 5000 scanned data were collected and averaged for each sample. The detailed carbon assignments and calculation of the copolymer composition followed the Randall’s method.27
The polymers were analyzed by differential scanning calorimetry (DSC) under nitrogen atmosphere with TA Q200 thermal analyzer. 8.0-10.0 mg sample was heated to 220 ℃ at 10 ℃ min-1, kept for 5 minutes at 220 ℃ to eliminate the thermal history, and then cooled down to -90 ℃ at 10 ℃ min-1 for recrystallization. After 5 minutes of isotherm at -90 ℃, the sample was reheated from -90 ℃ to 220 ℃ at 10 ℃ min-1.
Fractionation of Polymer. About 2 g polymer sample was completely dissolved in 200 mL boiling n-octane, where n-octane contained 0.1 wt% butylated hydroxytoluene (BHT) antioxidant. Then the solution was gradually cooled down to room temperature for 12 hours and precipitated. The insoluble part was separated from the solution by centrifugation. The part dissolved in n-octane was recovered from the solution by rotating evaporation, which was called Fraction A. The insoluble part was further extracted by 250 mL boiling n-heptane (containing 0.1 wt% BHT) in a Soxhlet extractor for 12 hours. It was further fractionated into two parts, namely soluble part (Fraction B) and insoluble part (Fraction C).
|
Figure 1 Diagrammatic sketch of experimental devices.26 |
|
Figure 2 Atmosphere composition control process. |
Gas-Phase Copolymerization Behavior of Ethylene, Propylene and Hydrogen under Controlled Atmosphere Composition. Firstly, run 1 (Table 1) was carried out at the same experimental conditions as the copolymerization run (run 7) reported in our previous article,26 whereas gases were fed in Mode II in run 1 and in Mode I in run 7.
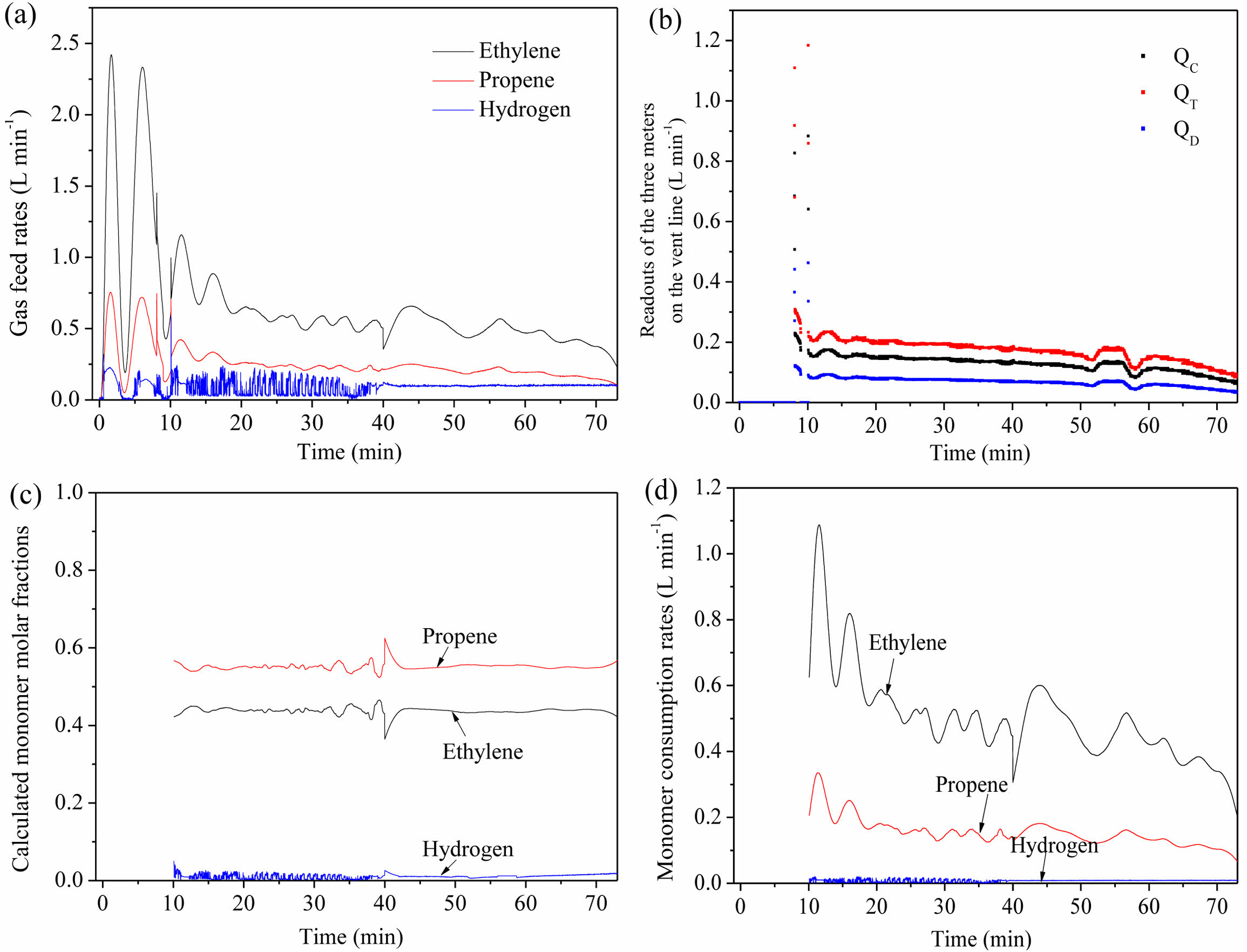
Figure 3(c) shows the atmosphere composition in the reactor during gas-phase copolymerization in run 1, which were calculated from the volume flow readings of the
Coriolis flowmeter, thermal flowmeter and differential pressure flowmeter (QC, QT and QD, Figure 3(b)) according to Equations (1-3). It can be seen that the atmosphere compositions were maintained at a near constant level, which verified the feasibility of atmosphere composition control process. The feed rates of monomers are available (monitored by DCS, Figure 3(a)), the volume flow of the exhaust equals the volume flow readings of the Coriolis flowmeter (QC). Therefore, the consumption rates of each monomer can be calculated according to the mass balance:

where Rp, Qin, Qout and y are the consumption rates, feed rates, vent rates and molar fractions of each monomer, V is the reactor volume (1 L) and t is the polymerization time. Since the molar fractions of monomer were almost unchanged during copolymerization, Equation (4) could be further simplified to:
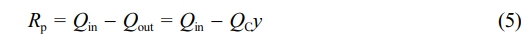
The derived consumption rates of monomers are shown in Figure 3(d). The decay trends of ethylene and propene consumption were similar, and hydrogen consumption was almost zero.
The atmosphere compositions in the reactor and monomer consumption rates during gas-phase copolymerization in run 7 were shown in Supporting Information (SI). The comparison between run 1 and run 7 illustrated that the polymerization kinetics (evolution trends of monomer consumption rates) varied with the atmosphere compositions in the reactor, which also resulted in differences on polymer properties.
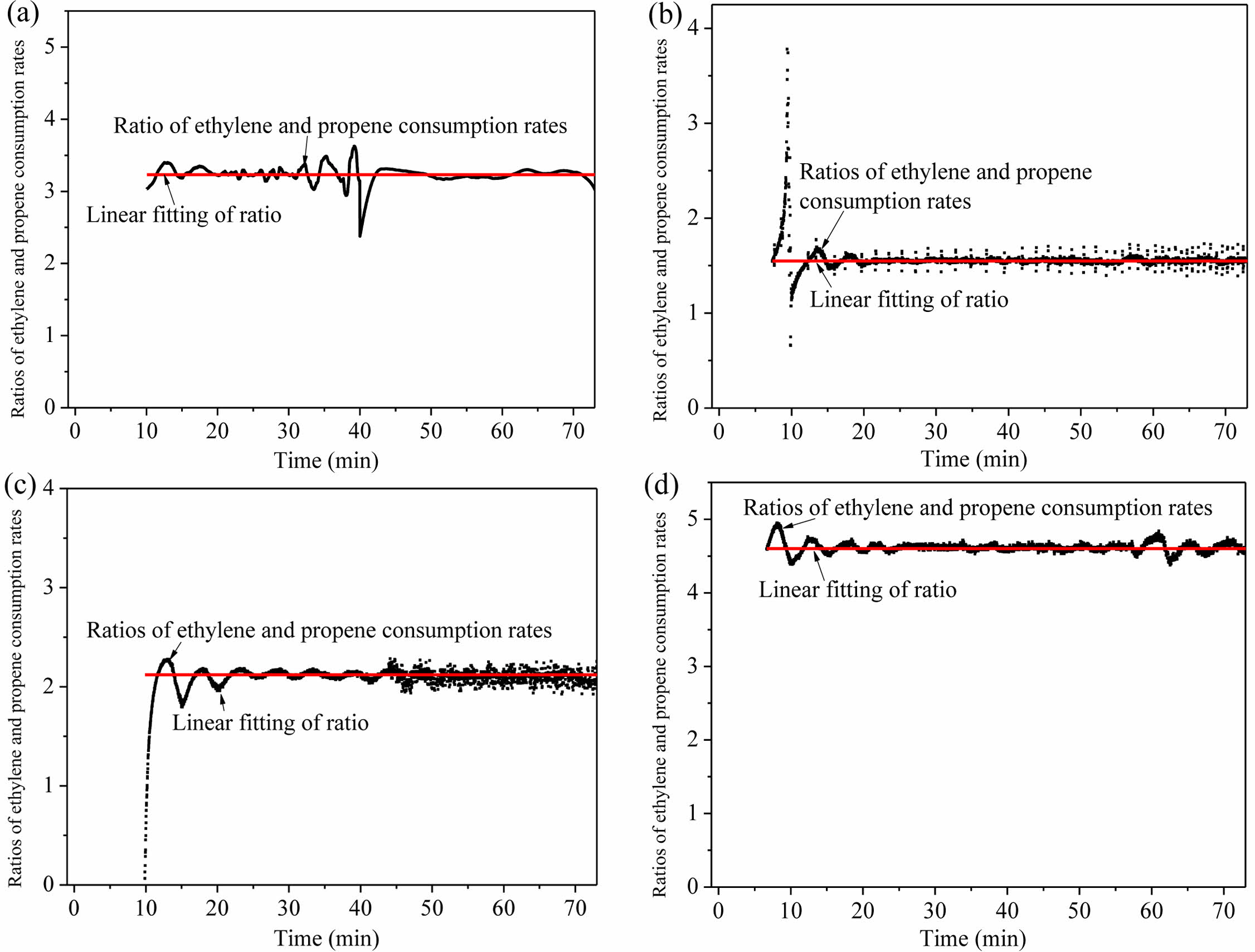
To further investigate the effect of atmosphere composition on ethylene/propene gas-phase copolymerization, runs with different atmosphere compositions in the reactor (runs 2-4, Table 1) were performed. In runs 2, 3, 1, and 4, the gases were fed in Mode II and the ethylene content in the controlled atmosphere increased. Based on the similar calculation, the atmosphere composition in the reactor and monomer consumption rates during gas-phase copolymerization in runs 2-4 were deduced (seen in SI). Similarly, the consumption rates of ethylene and propylene showed similar decay trends in each run. It was found that the ratios of ethylene to propylene consumption remained almost unchanged during each copolymerization process (Figure 4).
Because propene slurry polymerization in all runs were carried out under the same polymerization conditions (the repeatability has been confirmed in previous work26),
the results of polymerization were considered to be the same, that is, the polymer yield in propene slurry polymerization was considered to be 14.3 g. Then the polymer yield and copolymer composition of the products in gas-phase copolymerization in each run were calculated (Table 2).
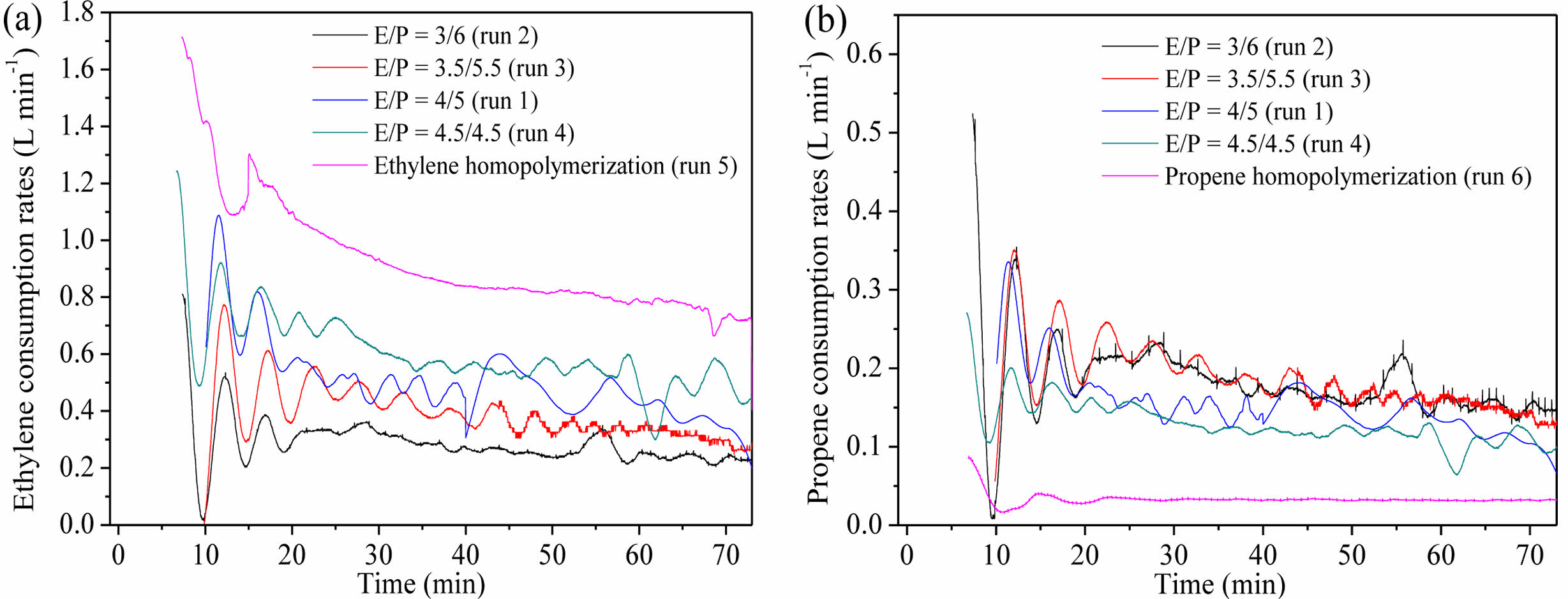
With the increase of ethylene content in the gas phase, polymer yield in copolymerization stage (II_Pro.) increased, while propene incorporation in the products in copolymerization stage (II_F͞͞P) decreased. The calculated II_F͞͞P values by the polymer yield and the molar fraction of propene in PP/P(E-co-P) were close to the ones calculated by the integration of ethylene and propene consumption during copolymerization, which verified the reliability of the atmosphere composition control process. As shown in Figure 5, ethylene consumption increased with the increase of ethylene content in the gas phase, while propene consumption decreased slightly. The ethylene consumption rates in copolymerization runs were lower than those in homopolymerization run (run 5), while the propene consumption rates in copolymerization runs were higher than those in homopolymerization run (run 6), which might be attributed to the “comonomer effect” (steric hindrance caused by the incorporation of bulkier propene).
Characterization and Fractionation of Polymer. In runs 1-4 and 6, the molecular weights of the polymers obtained were slightly higher than 200000 g/mol, and PDI increased with the increase of the ethylene content in the gas phase. The polymer with the highest molecular weight and the lowest PDI were prepared in run 5, which indicated that the polymer produced in copolymerization stage in run 5 had the highest molecular weight in all the polymers produced in copolymerization stage in all runs.
The polymer obtained in run 6 showed one melting peak with Tmof 163.28 ℃. The polymer obtained in run 5 showed two melting peaks, one around 135.38 ℃ (Tmof PE) and the other around 160.55 ℃ (Tmof PP). The area of the right peak is very small because the polypropylene produced in the first stage accounts for a small proportion of the polymer produced. The polymers obtained in runs 1-4 had two melting peaks, one around 120 ℃ and the other around 160 ℃. According to the sequence distribution of the polymers (Table 3) by 13C NMR spectra, the long ethylene or propene segments (EEE or PPP) in the ethylene/propylene copolymer may contribute to the left peak (Tmat about 120 ℃). The melting enthalpy (ΔH) and glass transition temperatures (Tg) of the polymers obtained in runs 1-4 were much lower than those in run 6, indicating that the elastomer component was produced during gas-phase copolymerization stage.
Table 4 shows the fractionation results of the polymer samples in runs 1-4. As shown, the weight percentages of Fractions A of all samples were more than 50%. Generally speaking, with the increase of ethylene content in the gas phase, the content of Fraction A increased, while the content of Fraction C decreased. For PP/P(E-co-P), Fraction A is mainly random ethylene/propene copolymer, Fraction B is segmented ethylene/propene copolymer,28 and Fraction C is basically highly isotactic PP.29 SI gives the GPC, DSC, Successive Self-nucleation and Annealing (SSA) and 13C NMR analysis of Fractions A-C.
Estimation of Reactivity Ratios of Ethylene and Propene. Firstly, Mayo-Lewis equation22 was used to evaluate the monomer reactivity ratios in ethylene/propene gas-phase copolymerization. The equation expresses the relationship between the ratio of monomer concentrations in feed (MA/MB) and the instantaneous molar ratio of monomers in copolymer (dMA/dMB, i.e. the ratio of monomer consumption rates):
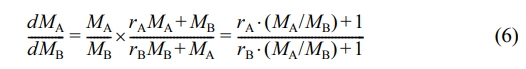
where
MAand MB represent the concentrations of ethylene and propene, and rA and rB represent the reactivity ratios of ethylene and propene. For semicrystalline PP/P(E-co-P), monomer sorption occurs only in the amorphous polymer phase under gas-phase condition. Under low polymerization pressure, the actual monomer concentrations can be determined according to Henry’s law:

where cm is the actual monomer concentration in the polymer, k is the Henry constant, and P* is the monomer partial pressure. The value of the Henry constant can be calculated from the following equation:30
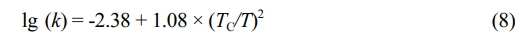
where TC denotes the critical temperature and k denotes the Henry constant in mol/(L·atm). The critical temperatures of ethylene and propene are 282.34 K and 364.76 K, respectively. Then, the Henry constants are 0.03784 mol/(L·atm) and 0.1217 mol/(L·atm), respectively. The atmosphere compositions remained almost unchanged during copolymerization, so the monomer partial pressures remained basically unchanged, and the monomer concentrations were calculated (Table 5).
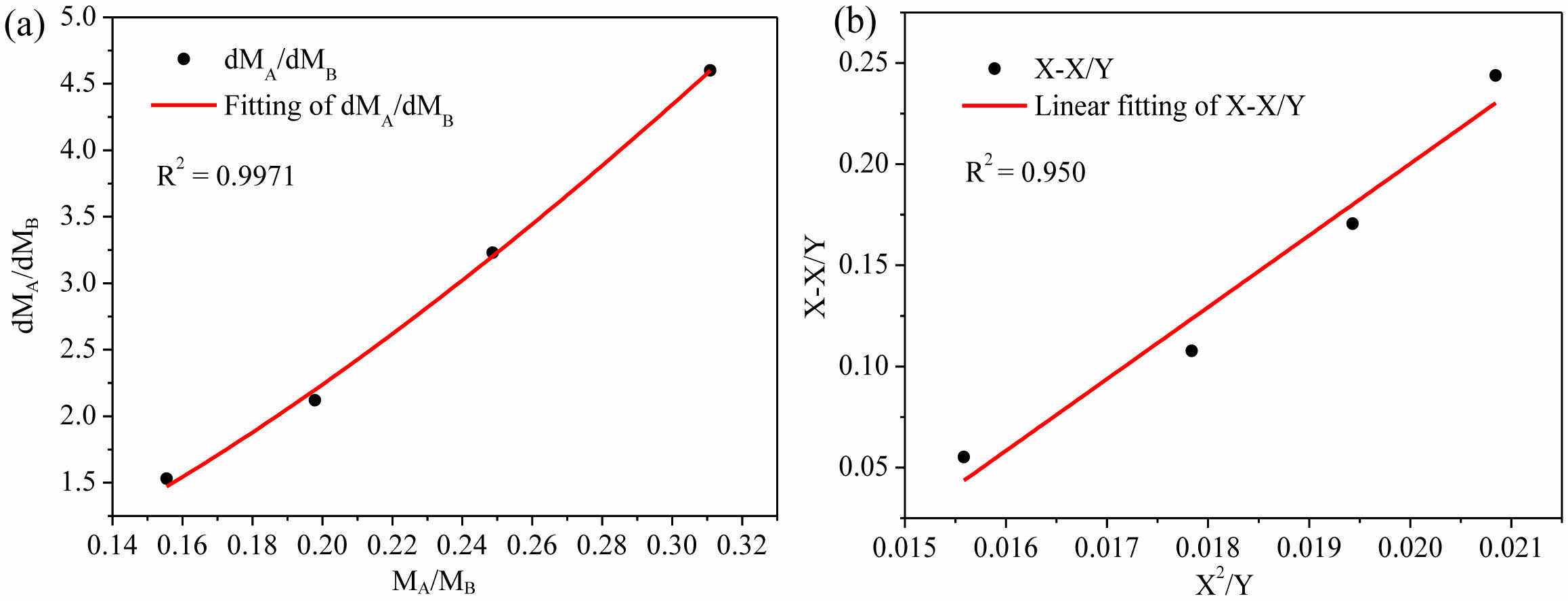
The ratios of monomer consumption rates (dMA/dMB) remained unchanged, their values were determined by linear fitting (Figure 4, Table 5), and then the monomer reactivity ratios were estimated according to Mayo-Lewis equation (Figure 6(a)). Good fitting was obtained and the calculated values with 95% confidence were rA= 42.28 ± 7.15, rB= 0.6452 ± 0.1419 and rArB = 27.28. The reactivity ratios were also evaluated by Fineman-Ross equation:31

where X and Y denote the ratio of monomer concentrations in feed (MA/MB) and the molar ratio of monomers in copolymer (calculated by II_F͞͞P in Table 2), respectively. The monomer reactivity ratios were estimated by linear fitting (Figure 6(b)), the values with 95% confidence were rA= 35.48 ± 4.65, rB= 0.5094 ± 0.0862 and rArB = 18.07. The reactivity ratios estimated by these two equations were close, rA> rB indicated that ethylene insertion was preferred, and rArB> 1 indicated the blockiness of the polymer chains produced in copolymerization stage. It's worth mentioning that gas-phase polymerization differs significantly from solution polymerization in several crucial aspects, and these distinctions have an impact on the
reactivity ratios of the monomers involved. In gas-phase polymerization, the monomers react with the catalyst surface in the vapor phase, resulting in surface-controlled kinetics due to restricted diffusion. The absence of a solvent eliminates solvent-induced chain transfer reactions, leading to cleaner and more predictable kinetic processes.
Nonetheless, temperature gradients within gas-phase reactors, particularly in fluidized beds, may introduce variations in the reactivity ratios of the monomers. Conversely, solution polymerization boasts higher monomer concentrations, superior temperature control, and solvent effects that influence reactivity and selectivity, which are not present in gas-phase processes.
|
Figure 3 (a) Gas feed rates; (b) volume flow readings of the coriolis flowmeter, thermal flowmeter and differential pressure flowmeter; (c) atmosphere composition in the reactor; (d) monomer consumption rates during gas-phase copolymerization in run 1. |
|
Figure 4 Ratios of ethylene to propene consumption during copolymerization in runs 1-4 (a-d). |
|
Figure 5 (a) Ethylene consumption rates in runs 1-5; (b) propene consumption rates in runs 1-4 and 6. |
|
Figure 6 Evaluation of monomer reactivity ratios using: (a) Mayo-Lewis equation; (b) Fineman-Ross equation. |
|
Table 1 Results of Ethylene/propene Gas-phase Copolymerization Under Controlled Atmosphere Compositions |
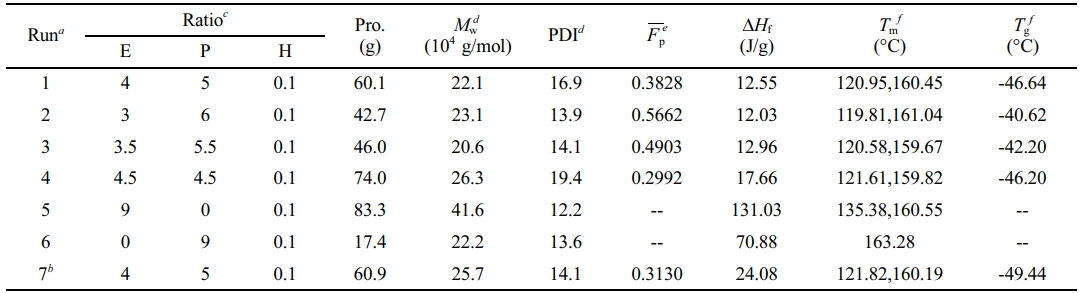
aExperimental conditions for preparing polypropylene particles: catalyst = 60 mg, TEA = 10 mL × 1 mol L-1 (Al/Ti = 138.9), C-Donor = 18 mL × 0.1 mol L-1 (Si/Ti = 25), n-hexane = 250 mL; propene pressure = 6 bar, polymerization temperature = 40 ℃, stir rate = 300 rpm, polymerization time = 30 min. Experimental conditions for gas-phase polymerization: The reduction valves on ethylene, propene and hydrogen intake lines were set as close as possible to 4 bar. The reduction valves on the vent line were set to 2.5 bar. DCS controlled the three flow controllers on the intake lines to feed ethylene, propene and hydrogen with constant atmosphere composition (monomer molar ratio) in the reactor (in Mode II) and the reactor pressure was kept at 3 bar. The Alicat flowmeter showed that the temperature of the exhaust was close to 30 ℃ and the pressure was kept at 1 atm. Polymerization temperature = 40 ℃, stir rate = 100 rpm, polymerization time = 72 min; bPreviously reported copolymerization run, where the gases were fed in Mode I26; cMolar ratio of ethylene, propene and hydrogen in the gas phase in the reactor; dMeasured by GPC; eMolar fraction of propene in polymer, measured by 13C NMR; fMeasured by DSC from the second heating curve. |
|
Table 2 Polymer Yields and Copolymer Compositions of the Products in Gas-phase Copolymerization in Runs 1-4 |
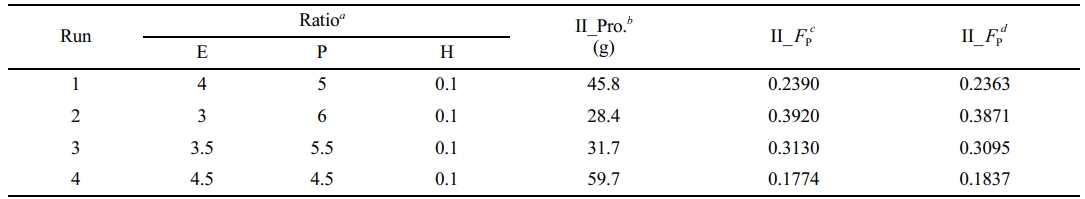
aMolar ratio of ethylene, propene and hydrogen in Mode II; bPolymer yield in copolymerization stage, calculated according to the polymer yield in homopolymerization stage (14.3 g) and the polymer yield in two-stage sequential polymerization; cMolar fraction of propene in the products in copolymerization stage, calculated according to the polymer yield and the molar fraction of propene in PP/P(E-co-P); dMolar fraction of propene in the products in copolymerization stage, calculated by integral calculation of the consumption rates of ethylene and propylene during copolymerization. |
|
Table 4 Fractionation Results of the Polymers Obtained in Runs 1-4 |
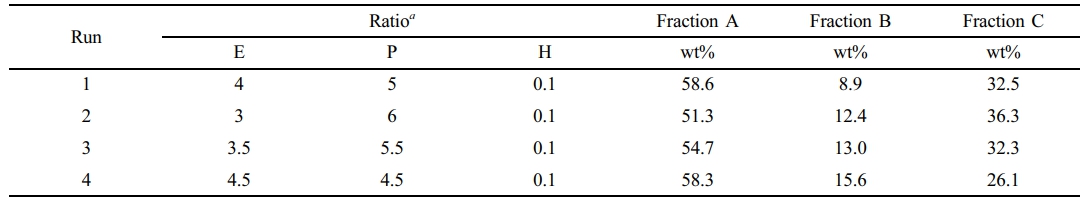
aMolar ratio of ethylene, propene and hydrogen in the atmosphere in the reactor. |
|
Table 5 Monomer Concentration and Ratio of Monomer Consumption Rates |
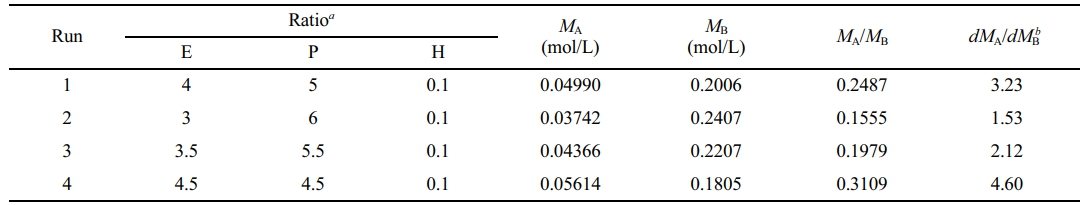
aMolar ratio of ethylene, propene and hydrogen in the atmosphere in the reactor; bDetermined by linear fitting of the ratios of ethylene and propene consumption rates (Figure 4). |
In this work, the method for detecting the atmosphere composition in the previous work26 was further developed to control the atmosphere composition during gas-phase copolymerization of ethylene, propene and hydrogen. During preparation of PP/P(E-co-P) in-reactor alloy, ethylene/propene gas-phase copolymerization with hydrogen modulation was performed. By the atmosphere composition control process, the atmosphere compositions were basically kept constant and the ratios of ethylene to propene consumption were also kept unchanged. With the increase of ethylene content in the gas phase, ethylene consumption rates increased, propene consumption rates decreased slightly, and propylene contents in the copolymers decreased. The structural heterogeneity of the polymers were characterized by GPC, 13C NMR, DSC analysis and solvent fractionation. The reactivity ratios of ethylene and propene were estimated by Mayo-Lewis equation and Fineman-Ross equation. The calculated values by the two equations were close.
- 1. Covezzi, M. The Spherilene Process: Linear Polyethylenes. Macromol. Symp.1995, 89, 577-586.
-

- 2. Reichert, K. H.; Moritz, H. U. 6th International Workshop on Polymer Reaction Engineering: Papers of the 6th International Workshop on Polymer Reaction Engineering, Berlin, Germany, 1998.
- 3. Ning, Y. N.; Dong, C. M.; Wang, W. Z.; Xu, H. Y.; Jiang, T. Recent Progress in Gas Phase Polypropylene Process. Chem. Ind. Eng. Prog. 2010, 29, 2220-2227.
- 4. Pasquini, N.; Addeo, A. Polypropylene Handbook; Hanser: Munich, 2005.
- 5. Covezzi, M.; Mei, G. The Multizone Circulating Reactor Technology. Chem.Eng. Sci. 2001, 56, 4059-4067.
-

- 6. Xu, Z. G.; Chakravarti, S.; Ray, W. H. Kinetic Study of Olefin Polymerization with a Supported Metallocene Catalyst. I. Ethylene/Propylene Copolymerization in Gas Phase. J. Appl. Polym. Sci. 2001, 80, 81-114.
-

- 7. Sinclair, K. B. 1985 Annual Meeting of the American Institute of Chemical Engineers: Papers of the 1985 Annual Meeting of the American Institute of Chemical Engineers, New York, America, 1985.
- 8. Mannan, T. M.; Hammawa, H.; Lynch, D. T.; Wanke, S. E. A Laboratory Reactor for Gas-Phase Olefin Polymerization. Can. J. Chem. Eng. 2004, 82, 371-381.
-

- 9. Cui, N. N.; Ke, Y. C.; Lu, Z. X.; Wu, C. H.; Hu, Y. L. Structure and Properties of Polypropylene Alloy In Situ Blends. J. Appl. Polym. Sci. 2006, 100, 4804-4810.
-

- 10. Fan, Z. Q.; Deng, J.; Zuo, Y. M.; Fu, Z. S. Influence of Copolymerization Conditions on the Structure and Properties of Polyethylene/Polypropylene/Poly(ethylene-co-propylene) In-Reactor Alloys Synthesized in Gas-Phase with Spherical Ziegler-Natta Catalyst. J. Appl. Polym. Sci. 2006, 102, 2481-2487.
-

- 11. Zohuri, G. H.; Sadegvandi, F.; Jamjah, R.; Ahmadjo, S.; Nekoomanesh, M.; Bigdelli, E. Copolymerization of Ethylene/Propylene Elastomer Using High-Activity Ziegler-Natta Catalyst System of MgCl2 (Ethoxide Type)/EB/PDMS/TiCl4/PMT. J. Appl. Polym. Sci. 2002, 84, 785-790.
-

- 12. Zohuri, G.; Mortazavi, M.; Jamjah, R.; Ahmadjo, S. Copolymerization of Ethylene-Propylene Using High-Activity Bi-Supported Ziegler-Natta TiCl4 Catalyst. J. Appl. Polym. Sci. 2004, 93, 2597-2605.
-

- 13. Kou, B.; Mcauley, K. B.; Hsu, C. C.; Bacon, D. W.; Yao, K. Z. Gas-Phase Ethylene/Hexene Copolymerization with Metallocene Catalyst in a Laboratory-Scale Reactor. Ind. Eng. Chem. Res. 2005, 44, 2443-2450.
-

- 14. Mehtarani, R.; Fu, Z. S.; Tu, S. T.; Fan, Z. Q.; Tian, Z.; Feng, L. F. Synthesis of Polypropylene/Poly(ethylene-co-propylene) In-Reactor Alloys by Periodic Switching Polymerization Process: Dynamic Change of Gas-Phase Monomer Composition and Its Influences on Polymer Structure and Properties. Ind. Eng. Chem. Res. 2013, 52, 9775-9782.
-

- 15. Tan, N. Effect of Temperature on Property Homo- and Co-Polymerization Kinetics and Microstructure of Polymer Made with Ziegler-Natta Catalyst. Ph.D. Thesis, Beijing University of Chemical Technology, China, 2015.
- 16. Lynch, D. T.; Wanke, S. E. Reactor Design and Operation for Gas-Phase Ethylene Polymerization Using Ziegler-Natta Catalysts. Can. J. Chem. Eng. 1991, 69, 332- 339.
-

- 17. Chen, C. M. Gas Phase Olefin Copolymerization with Ziegler-Natta Catalysts. Ph.D. Thesis, University of Wisconsin-Madison, America, 1993.
- 18. Debling, J. A. Modeling Particle Growth and Morphology of Impact Polypropylene Produced in the Gas Phase. Ph.D. Thesis, University of Wisconsin-Madison, America, 1997.
- 19. Han-Adebekun, G. C. Heterogeneous Catalyzed Gas Phase Polymerization of Olefins: Kinetic Studies and Modeling Investigations. Ph.D. Thesis, University of Wisconsin-Madison, America, 1997.
- 20. Bergstra, M. F.; Weickert, G. Semi-Batch Reactor for Kinetic Measurements of Catalyzed Olefin Co-Polymerizations in Gas and Slurry Phase. Chem. Eng. Sci. 2006, 61, 4909-4918.
-

- 21. Bergstra, M. F.; Weickert, G.; Meier, G. B. Metallocene-Catalyzed Gas-Phase Ethylene Copolymerization: Kinetics and Polymer Properties. Macromol. React. Eng. 2009, 3, 433-447.
-

- 22. Mayo, F. R.; Lewis, F. M.; Walling, C. Copolymerization. VIII. The Relation Between Structure and Reactivity of Monomers in Copolymerization. J. Am. Chem. Soc. 1948, 70, 1529-1533.
-

- 23. Liu, W. F.; Zhang, K. L.; Fan, H.; Wang, W. J.; Li, B. G.; Zhu, S. P. Living Copolymerization of Ethylene/1-Octene with Fluorinated FI-Ti Catalyst. J. Polym. Sci. Part A: Polym. Chem. 2012, 51, 405-414.
-

- 24. Liu, W. F.; Guo, S.; Fan, H.; Wang, W. J.; Li, B. G. Synthesis of Ethylene/1-Octene Copolymers with Controlled Block Structures by Semibatch Living Copolymerization. AIChE J. 2013, 59, 4686-4695.
-

- 25. Han-Adebekun, G. C.; Hamba, M.; Ray, W. H. Kinetic Study of Gas Phase Olefin Polymerization with a TiCl4/MgCl2 Catalyst I. Effect of Polymerization Conditions. J. Polym. Sci. Part A: Polym. Chem. 1997, 35(10), 2063-2074.
-

- 26. Zheng, Z.; Yang, Y. L.; Huang, K.; Hu, J. J.; Jie, S. Y.; Li, B. G. Real-Time Detection of Atmosphere Composition in Three-Component Gas-Phase Copolymerization of Olefins. Macromol. React. Eng. 2018, 12, 1800042.
-

- 27. Randall, J. C.; Hsieh, E. T. Carbon-13 NMR Tetrad Assignments in Ethylene- Propylene Copolymers. Macromolecules 1982, 15, 1584-1586.
-

- 28. Fu, Z. S.; Tu, S. T.; Fan, Z. Q. Effect of the Combined External Electron Donors on the Structure and Properties of Polypropylene/Poly(ethylene-co-propylene) In-Reactor Alloys Prepared by High-Efficiency Industrial Ziegler-Natta Catalyst. Ind.Eng. Chem. Res. 2013, 52, 5887-5894.
-

- 29. Fan, Z. Q.; Zhang, Y. Q.; Xu, J. T.; Wang, H. T.; Feng, L. X. Structure and Properties of Polypropylene/poly(ethylene-co-propylene) In-situ Blends Synthesized by Spherical Ziegler-Natta catalyst. Polymer 2001, 42, 5559-5566.
-

- 30. Hutchinson, R. A.; Ray, W. H. Polymerization of Olefins Through Heterogeneous Catalysis. VIII. Monomer Sorption Effects. J. Appl. Polym. Sci. 1990, 41, 51-81.
-

- 31. Fineman, M.; Ross, S. D. Linear Method for Determining Monomer Reactivity Ratios in Copolymerization. J. Polym. Sci. 1950, 5, 259-262.
-

- Polymer(Korea) 폴리머
- Frequency : Bimonthly(odd)
ISSN 2234-8077(Online)
Abbr. Polym. Korea - 2024 Impact Factor : 0.6
- Indexed in SCIE
 This Article
This Article
-
2025; 49(2): 187-196
Published online Mar 25, 2025
- 10.7317/pk.2025.49.2.187
- Received on Sep 10, 2024
- Revised on Nov 26, 2024
- Accepted on Dec 6, 2024
 Services
Services
- Full Text PDF
- Abstract
- ToC
- Acknowledgements
- Conflict of Interest
- Supporting Information
Introduction
Experimental
Results and Discussion
Conclusions
- References
Shared
 Correspondence to
Correspondence to
- Zheng Zheng
-
Zhejiang Industrial Polytechnic College, 151 Qutun Road, Yuecheng District, Shaoxing, Zhejiang 312000, China
- E-mail: 20240017@zjipc.edu.cn
- ORCID:
0000-0003-4843-9381









 Copyright(c) The Polymer Society of Korea. All right reserved.
Copyright(c) The Polymer Society of Korea. All right reserved.