- Evaluation of Ultraviolet Stability and Surface Properties of Poly(methyl methacrylate) Doped with Metal Oxide Nanoparticles
Dina S. Ahmed, Emad Yousif*, Mohammed Kadhom**, Ahmed Ahmed***, Amani A. Husain***, Hassan Hashim****, Tayser S. Gaaz*****, Muna Bufaroosha******, Benson M. Kariuki*******, and Gamal A. El-Hiti********,†

Department of Chemical Industries, Institute of Technology-Baghdad, Middle Technical University, Baghdad, 10074 Iraq
*Department of Chemistry, College of Science, Al-Nahrain University, Baghdad, 64021 Iraq
**Department of Environmental Science, College of Energy and Environmental Science, Al-Karkh University of Science, Baghdad, 10081 Iraq
***Polymer Research Unit, College of Science, Al-Mustansiriyah University, Baghdad, 10052 Iraq
****Department of Physics, College of Science, Al-Nahrain University, Baghdad, 64021 Iraq
*****Air Conditioning and Refrigeration Techniques Engineering Department, College of Engineering and Technologies, Al-Mustaqbal University, Babylon, 10052 Iraq
******Department of Chemistry, College of Science, United Arab Emirates University, Al-Ain 1818, United Arab Emirates
*******School of Chemistry, Cardiff University, Main Building, Park Place, Cardiff, CF10 3AT, UK
********Department of Optometry, College of Applied Medical Sciences, King Saud University, Riyadh 11433, Saudi Arabia- 금속 산화물 나노 입자가 도핑된 폴리(메틸 메타크릴레이트)의 자외선 안정성 및 표면 특성 평가
Reproduction, stored in a retrieval system, or transmitted in any form of any part of this publication is permitted only by written permission from the Polymer Society of Korea.
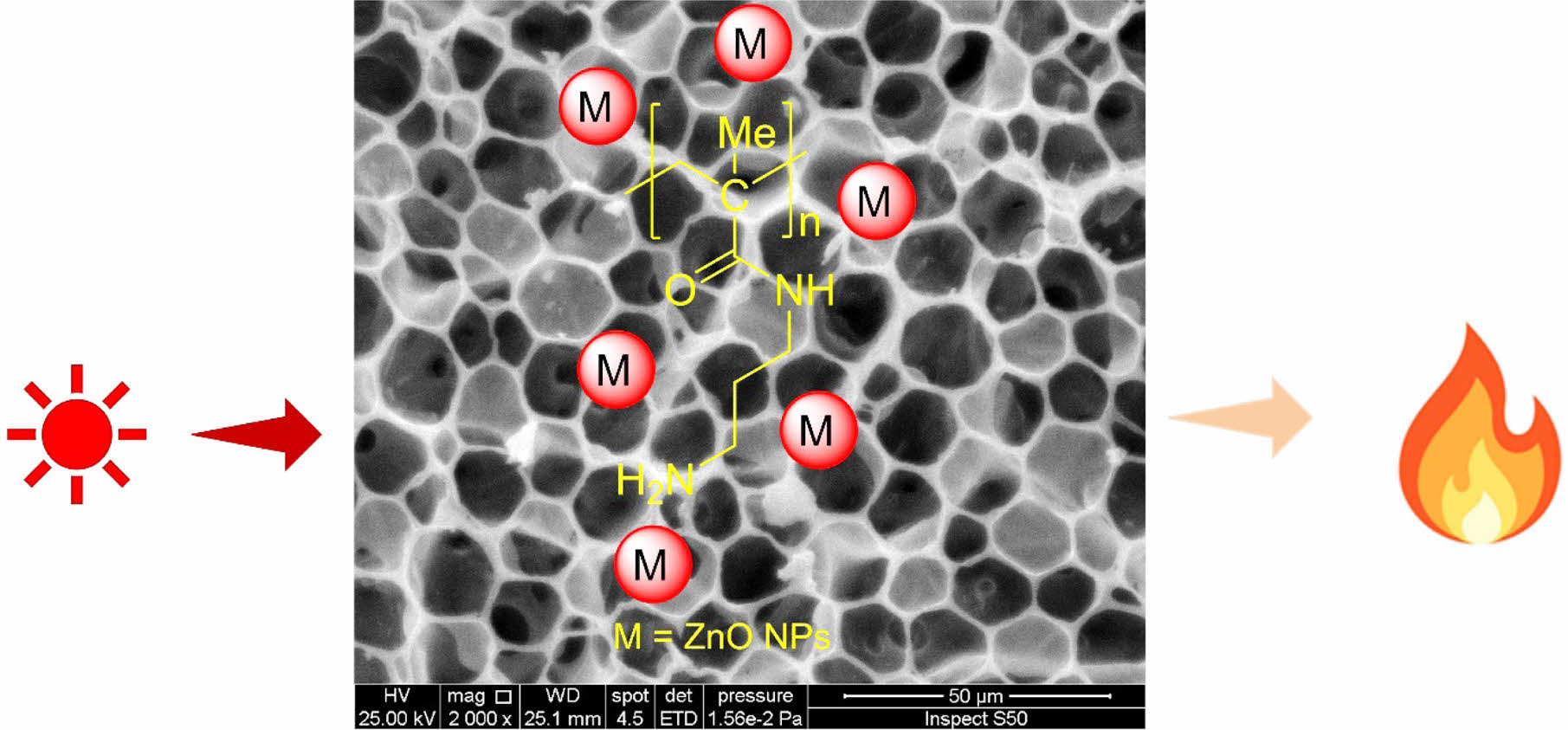
Photostabilizing polymeric materials is crucial for protecting them from damage by UV irradiation. Recent advancements have significantly enhanced polymer resistance to photooxidation and harsh environmental conditions by developing polymeric additives designed to act as photostabilizers. The present study assesses the significant impact of metal oxide nanoparticles (NPs) on enhancing the surface properties of poly(methyl methacrylate) (PMMA) and paving the way for more durable applications. The reaction of PMMA and propylenediamine led to the incorporation of amino residues, which was followed by the attachment of various metal oxide NPs, namely nickel oxide (NiO), titanium dioxide (TiO2), magnesium oxide (MgO), and zinc oxide (ZnO). Thin PMMA films doped with metal oxide NPs experienced reduced photodegradation compared to PMMA films containing the amino residues only. Of the metal oxide NPs studied, PMMA doped with ZnO NPs exhibited the lowest level of weight loss and surface damage caused by UV irradiation. These findings indicate the potential of metal oxide NPs in enhancing the photostability and surface properties of PMMA, contributing to the development of more durable polymeric materials.
The surface of poly(methyl methacrylate) was chemically modified effectively by incorporating metal oxide nanoparticles. The modification enables poly(methyl methacrylate) to resist photodegradation when exposed to ultraviolet light. Poly(methyl methacrylate) containing zinc oxide nanoparticles showed the highest level of protection against photodegradation.
Keywords: surface modification, poly(methyl methacrylate), ultraviolet irradiation, surface morphology, metal oxide nanoparticles.
We thank Al-Nahrain University for its technical support. The authors acknowledge the support received from the Researchers Supporting Project (number RSP2024R404), King Saud University, Riyadh, Saudi Arabia.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Polymers are valued for their diverse properties, low cost, and moldability.1 Physical methods modify surfaces, while chemical processes attach functional groups to polymer chains. Mechanical methods create microstructures.2 Blending and doping with organic materials improve polymer performance, enhancing properties like corrosion resistance and stability.3 Continued research is crucial for advancing efficient polymer composites for various applications, considering factors like miscibility, dispersion, and particle size.4-7
Incorporating new materials using different processes, such as doping and blending, can greatly enhance the performance of basic polymeric materials. Such processes can significantly improve the morphological, structural, mechanical, and electrical properties of polymers, making them suitable for a wide range of applications.8-10 The quality of polymer blends can be enhanced by considering factors such as the compatibility of the components, phase separation, distribution of phases, particle size, interactions between the polymer molecules, and their crystalline arrangement.11 The capability of these polymers to blend can also be modified in the molten state.12,13 Organic conducting polymers with extended π-electron conjugation are an example of a class of materials of particular interest to researchers. These plastics have remarkable electrical properties, morphology, and crystallinity, which distinguish them from traditional insulating polymers.14,15
A variety of methods are employed in the fabrication of polymeric nanocomposite materials, showcasing the versatility of the research. These methods include ball milling, ex-situ and in-situ polymerization, plasma coating, injection molding, extrusion molding, and other techniques.11 Notably, in situ polymerization is exceptionally efficient in achieving a consistent distribution of fillers within the organic structure.16,17 Poly(methyl methacrylate) (PMMA) is a widely used polymer due to its excellent properties, including thermal stability, transparency, mechanical strength, and electrical insulation.18 The PMMA matrix can be enhanced by, for example, the addition of silica nanoparticles. Atom transfer radical polymerization ensures an even distribution of particles throughout the polymer matrix. Friction spot welding is a viable option for constructing nanocomposite engineering structures.19 Progress has been made in modifying polymers for various applications. However, research continues to address the limitations of current processes.
As part of ongoing research into using additives to enhance the stability of polymeric materials,20-26 we have previously shown that incorporating metal complexes containing naphthalene can effectively stabilize PMMA.27 The current study examines the surface modification of PMMA using propylene diamine and metal oxide NPs to significantly boost its photostability and enable more durable applications. Nickel oxide (NiO), titanium dioxide (TiO2), magnesium oxide (MgO), and zinc oxide (ZnO) NPs were added to the polymeric chains containing the amino residues. A range of techniques were used to characterize the materials, and the results indicated that the PMMA containing metal oxides, especially the one modified with ZnO NPs, degraded at a slower rate compared to the original polymer.
Materials and Methods. Materials: PMMA of a molar mass of 74315 g/mol, metal oxides (TiO2, ZnO, MgO, and NiO) of a purity of 97–98%, propylene diamine of a purity of 99%, and solvents of analytical grade were purchased from Merck (Gillingham, UK).
Preparation of PMMA Containing Propylenediamine: 5 g of PMMA was agitated in a 15 mL mixture of 2-propanol and water of a 1:1 ratio for 10 min at 25 ℃. The polymer was isolated and immersed in a solution containing 30 mg propylenediamine (H2N(CH2)3NH2) in 15 mL dimethyl sulfoxide (DMSO). The mixture was refluxed for 1 h while being continuously stirred. Following the removal of the solvent, the polymer was dried in air for 24 h at 25 ℃.
Synthesis of Modified PMMA Films Containing Metal Oxide NPS: To incorporate metal oxide NPs into the PMMA films, a mixture of 5 g of PMMA, 30 mg of propylene diamine, and 0.35 mg of the metal oxide nanoparticles was subjected to sonication in 100 mL of CHCl3 at 25 ℃ for 1 h. Subsequently, the blend was heated for 3 h with continuous stirring. The mixture was left to cool, followed by casting on a plate of glass containing holes with a thickness of 40 µm. The films were left to dry at 25 ℃ for 24 h, followed by 3 h in a vacuum oven at 40 ℃.
PMMA Films Irradiation: The PMMA films were exposed to UV irradiation (wavelength = 365 nm) utilizing a QUV accelerated weathering tester from Q-Panel Company in Homestead, FL, USA. The device was equipped with a stainless-steel plate and two 40 W UV fluorescent lamps (UV-B 365), which were positioned on opposite sides. The PMMA films underwent UV irradiation at a temperature of 25 ℃, with a light intensity of 6.2×10-9 Einstein /dm.s. The films were placed 10 cm from the tester in a vertical arrangement, aligned with the fluorescent bulbs. They were systematically rotated to provide uniform exposure.
FTIR Spectroscopy of PMMA Films: The Fourier transform infrared (FTIR) spectra were obtained using a Shimadzu FTIR-8300 spectrometer (Tokyo, Japan) to detect the increase in the absorption band intensity of the hydroxyl group (OH) throughout the irradiation process. The PMMA films underwent irradiation for various durations, ranging from 50 to 300 h in increments of 50 h. The enhanced intensity of the OH group peak at 3204 cm-1 was measured and compared with that of a reference peak at 1443 cm-1 (C–H), which did not change during irradiation. The hydroxyl group index (IOH) was determined for each irradiation time using Equation 1 from the intensities of the absorption bands of OH and the reference (As and Ar, respectively).28

Weight Loss of Irradiated PMMA Films: The decrease in the weight of the PMMA film was measured to evaluate the influence of irradiation on the photodegradation of the polymeric material. The weight loss percentage of the PMMA films was determined at variant irradiation times using Equation 2, based on the initial weight of the films (Wo) and the weight after irradiation (Wt).29
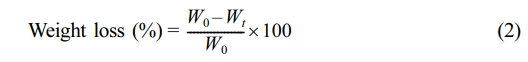
Surface Morphology of Irradiated PMMA Films. The surface damage of PMMA films due to irradiation was analyzed utilizing several microscopy techniques. Optical microscopy images were acquired using a Meiji Techno microscope (Tokyo, Japan). Scanning electron microscopy (SEM) and atomic force microscopy (AFM) images were obtained using a SIGMA 500 VP SEM (Carl Zeiss Microscopy, NY, USA) and a Veeco AFM (Plainview, NY, USA), respectively.
Surface Modifications of PMMA. A simple aminolysis process was employed to modify the PMMA, resulting in the introduction of an amine branch.30,31 This method is frequently used to add amine functionality to the PMMA chain surface. The process requires the reaction between PMMA and propylene diamine in DMSO and involves an SN2 reaction that generates an amino-functionalized PMMA surface. The amino-functionalized PMMA was doped with different metal oxide NPs, utilizing CHCl3 as the solvent (Scheme 1).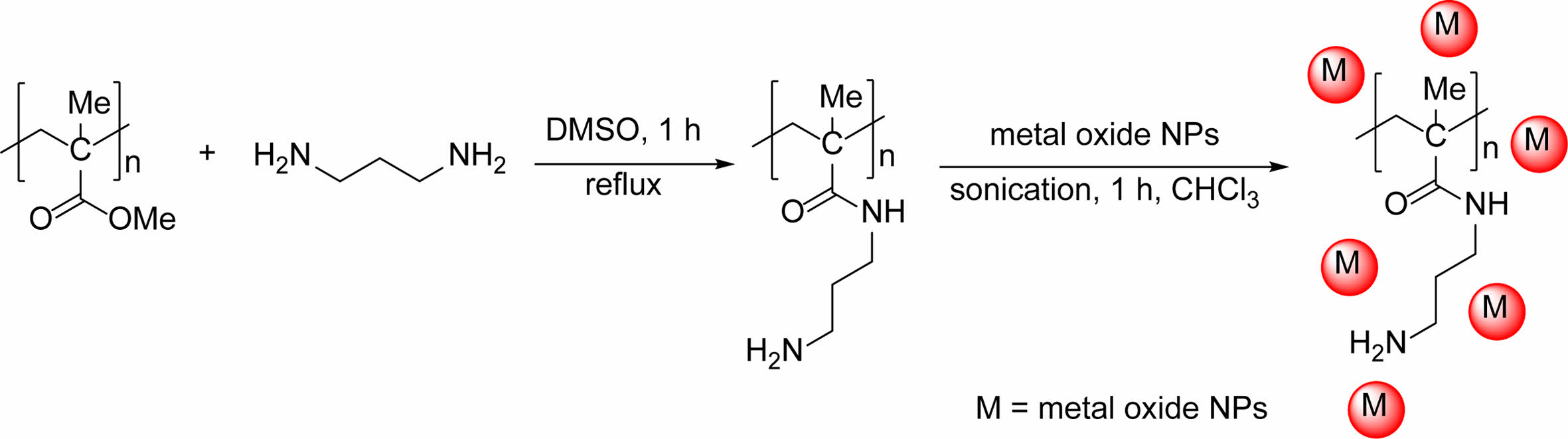
Scheme 1. Modification of PMMA by aminolysis process doped with different metal oxide NPs.
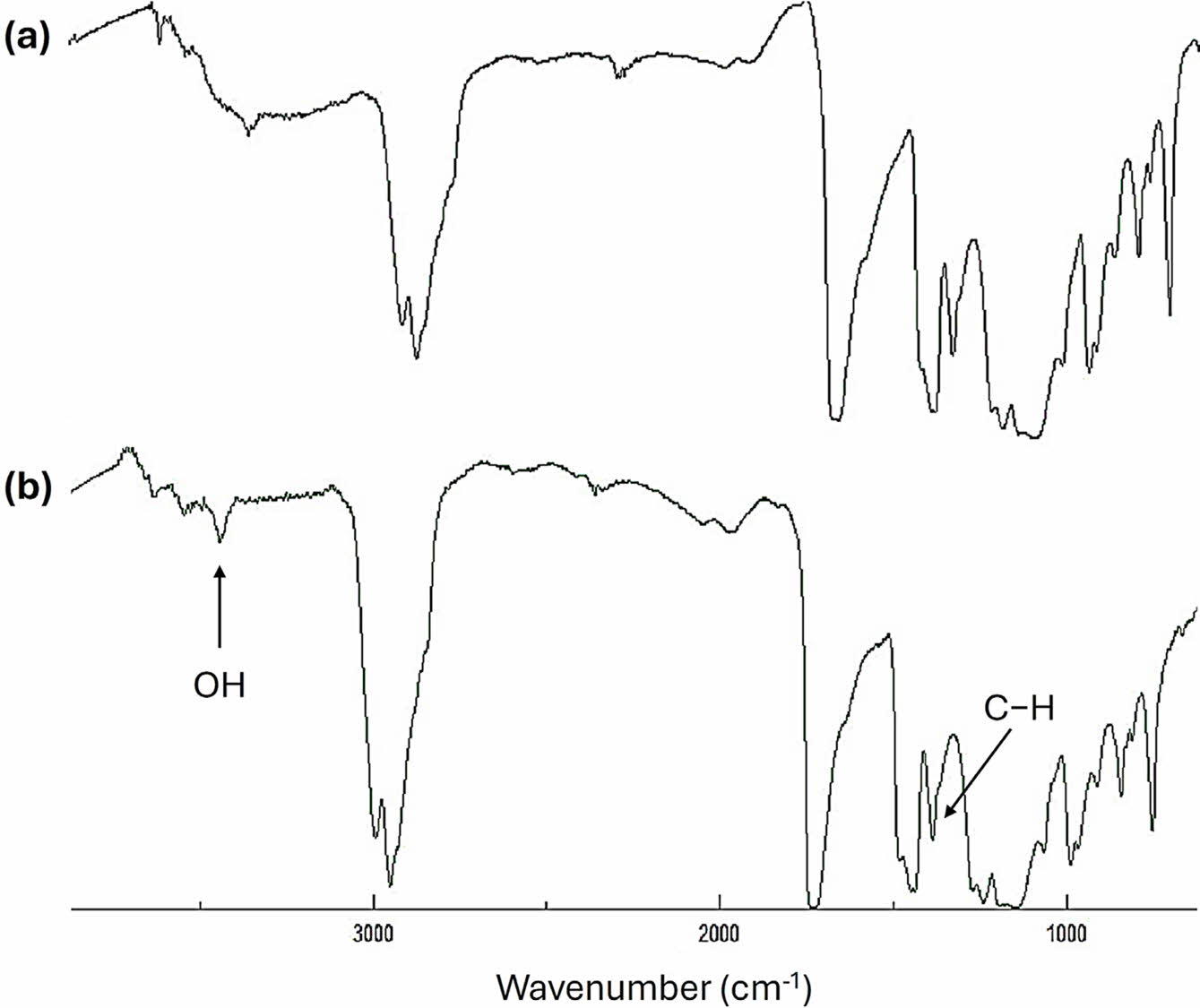
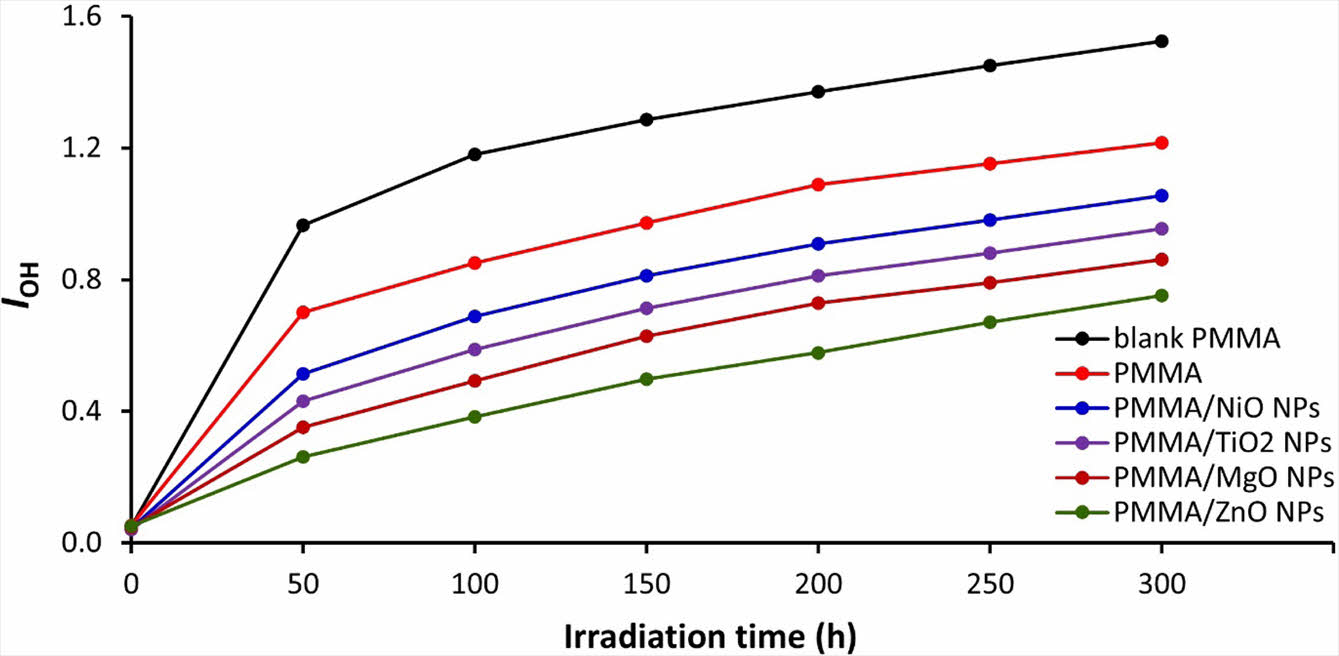
Examination of PMMA Films via FTIR Spectroscopy. The process of photochemical breakdown of PMMA results in the production of fragments that include distinct functional groups, including hydroxyl (OH) groups. Exposure of PMMA to UV light causes the C–Me and C–CO2Me bonds to undergo homolytic breakage, leading to the generation of free radicals. Free radicals combine with oxygen to generate peroxy radicals, which then extract hydrogen atoms from the PMMA polymeric chain and produce hydroperoxides (O–OH). The O–OH bonds undergo homolytic breakage, which results in the formation of alkoxy radicals within the polymer.32,33 The alkoxy radicals continue to remove hydrogen atoms and produce alcohols, which can be detected via FTIR spectroscopy. To quantify the extent of photodegradation, the PMMA films were exposed to UV radiation, and the alterations in the intensity of the OH absorption band at 3204 cm-1 in the FTIR spectra were observed and compared to the band at 1443 cm-1 due to the C–H group (Figure 1). The increase in the intensity of the OH absorption band was used as an indication of the degree of photodegradation in the PMMA.
The FTIR spectra of PMMA films revealed noticeable changes after irradiated with UV light. The emergence of the band at 3204 cm-1 is associated with the generation of OH groups. The increase in the hydroxyl group index (IOH) for aminated PMMA films with metal oxide NPs is gradual (Figure 2) but sharper at the beginning (over the first 50 h) of the irradiation process. The films containing metal oxide NPs have lower IOH values compared with the blank PMMA and the one that contains amino residue (aminated PMMA). ZnO NPs led to the lowest degradation and IOH. For example, after 300 h of irradiation, the IOH were 1.52, 1.22, 1.01, 0.86, 0.86, and 0.75 for the blank PMMA, aminated, PMMA/NiO NPs, PMMA/TiO2 NPs, PMMA/MgO NPs, and PMMA/ZnO NPs films, respectively. Metal oxide nanoparticles have properties that make them useful in a wide range of applications. The metals are acidic and can decompose active species, such as hydroperoxides, that cause the photodegradation of polymers. The most significant stabilizing effect was observed when ZnO NPs were used, but it is unclear why the PMMA film containing ZnO was more stable than the others. The particle size of metals affects the absorption of UV irradiation.34 However, UV light absorption is not the only factor that determines the efficiency of reducing PMMA photodegradation.
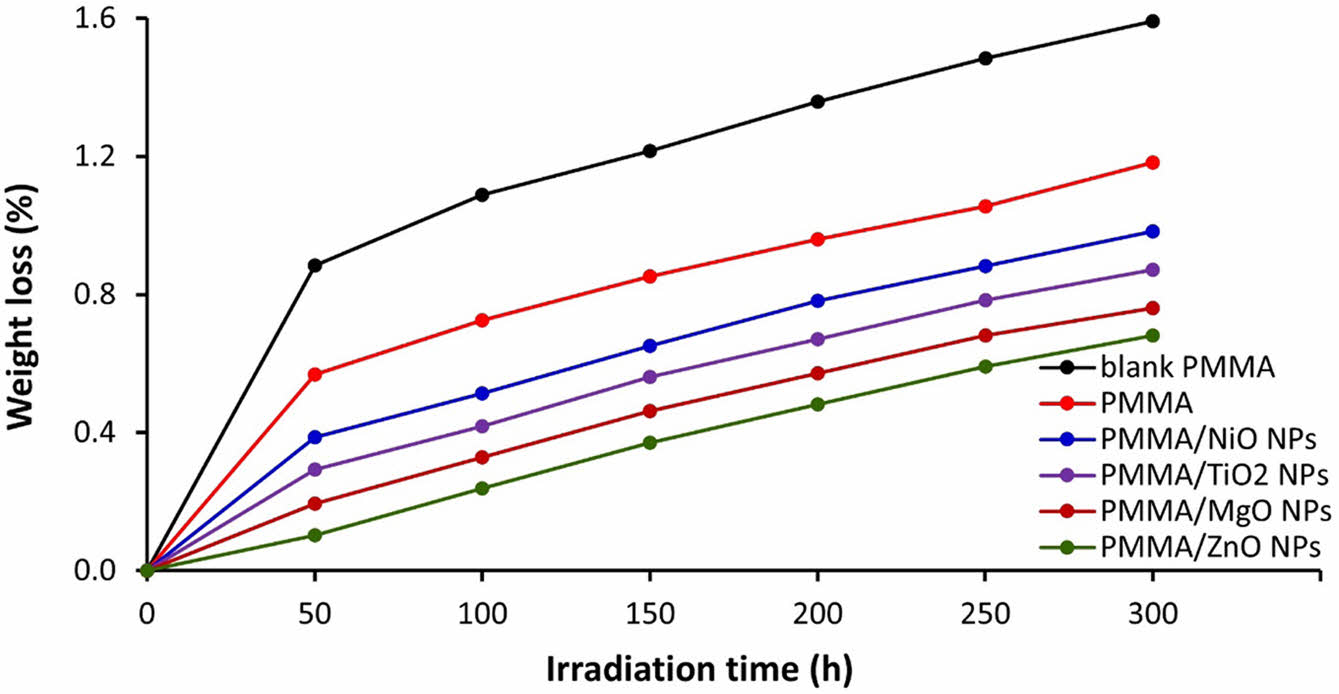
Determination of PMMA Films Weight Loss. Photodegradation of polymers results in bond cleavage and the elimination of low molecular weight fragments. Therefore, there is a noticeable weight loss after the photodegradation of polymers.35,36 The weight loss percentage is commonly acknowledged as a reliable indicator of the magnitude of photodegradation of polymeric materials due to photoirradiation. The weight loss (%) of PMMA films was determined every 50 h of the irradiation process and up to 300 h using Equation 2. The results obtained are shown in Figure 3, and they agree with those shown from FTIR (Figure 2). The PMMA containing ZnO NPs showed the most stability against irradiation, and the unmodified PMMA showed the most weight loss. The results are consistent with the incorporation of metal oxide NPs, which contribute to the protection of PMMA polymeric materials against UV radiation. At the end of the irradiation process, the weight losses (%) were 1.59, 1.18, 0.98, 0.87, 0.76, and 0.68 for the blank PMMA, aminated, PMMA/NiO NPs, PMMA/TiO2 NPs, PMMA/MgO NPs, and PMMA/ZnO NPs films, respectively.
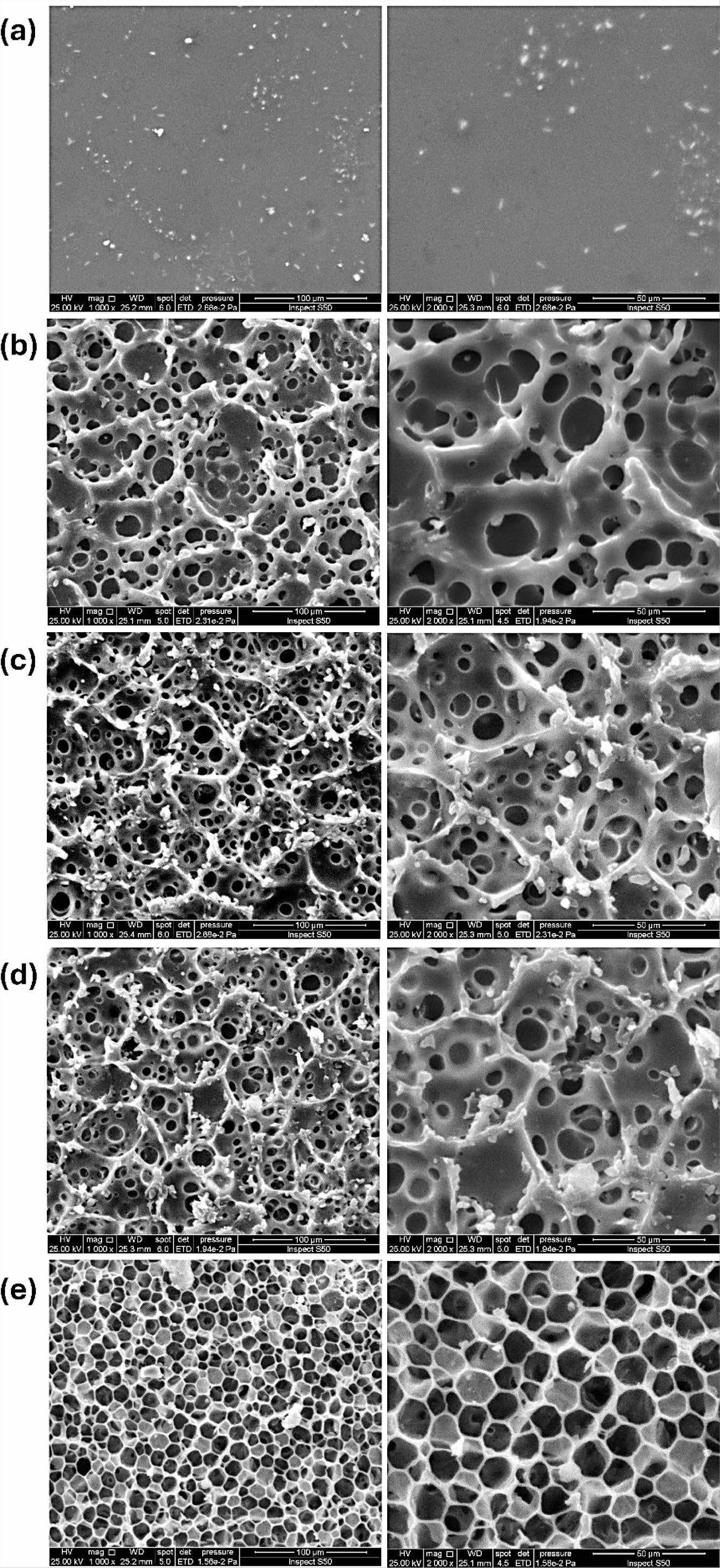
Surface Morphology Analysis. Various microscopic techniques can be used to analyze the surfaces of materials.37-41 SEM was employed to examine the aggregation, shapes, and sizes of polymer particles, as well as the degradation caused by irradiation of PMMA films. SEM images of PMMA films after irradiation are illustrated in Figures 4(a)–(e). Prior to irradiation, the polymer surface often exhibits a consistent and sleek appearance.40 The morphology of the chemically modified PMMA film (Figure 4(a)) exhibited a smooth texture without any visible gaps, fissures, or clustered particles, indicating a more compact structure. Photodegradation commonly causes weight reduction and agglomeration because of inadequate compatibility between the polymer and additives, leading to the appearance of voids.
The films containing metal oxide NPs generally exhibit a sponge-like structure (Figures 4(b)–(d)) with two distinct forms: an interconnected network type and a closed-cell type made of separate individual cells. An ideal asymmetric film features a thin outer layer with a network of interconnected pores. The high degree of interconnectedness within this porous substructure is vital for reducing resistance to mass flow, although measuring this characteristic might be difficult. However, this structure is preferred due to its superior mechanical properties compared to other configurations.42 The PMMA film doped with ZnO NPs exhibited a honeycomb-like architecture (Figure 4(e)). The coarse texture is most likely a result of the interaction or coordination between the PMMA-propylene diamine and ZnO NPs, presumably induced by cross-linking. It is an indication that the PMMA blend led to a homogeneous matrix. The surface had numerous circular pores, likely formed due to the fast evaporation of CHCl3, which was employed as the solvent during the fabrication of the film. The variation in pore size is likely attributable to discrepancies in the driving power that triggers phase separation.43 Similar observations have been made previously on the irradiation of polymeric materials.44-46
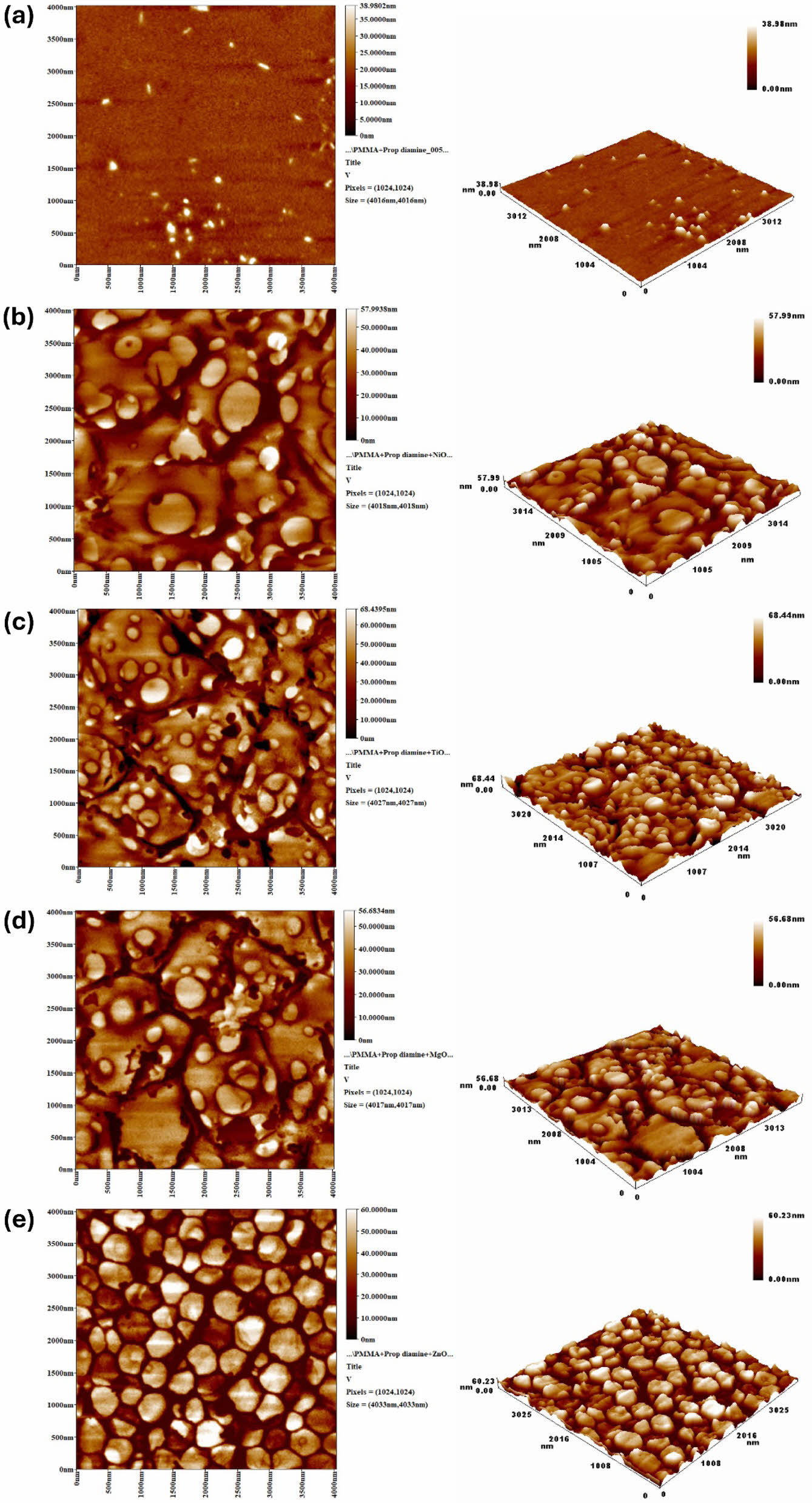
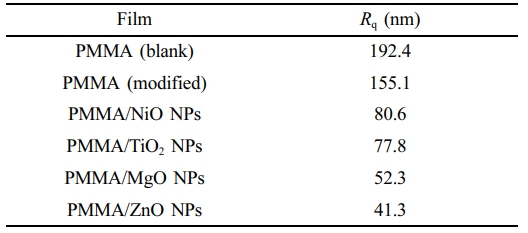
AFM is a versatile and straightforward method used in numerous disciplines to obtain two- and three-dimensional images of sample surfaces. Unlike other methods, AFM does not require a vacuum environment to conduct the test. Figure 5 illustrates the influence of irradiation on the morphological structure of PMMA films. The AFM images showed similar patterns to those observed in the SEM images shown in Figure 4. The images demonstrate that the surface of the modified PMMA film, which includes metal oxide NPs, maintained a high level of smoothness and exhibited minimal roughness following irradiation. The enhanced surface roughness in the unaltered PMMA film following irradiation may be attributed to the homogenous bond breakage.47,48 Surface roughness describes the irregularities present in a material. The roughness factor (Rq) is measured by the deviations from its ideal form along the normal vector. Significant deviations indicate roughness, while minimal ones suggest a smoother surface.49 Table 1 shows that the unmodified PMMA had the maximum roughness factor (Rq; nm) of 192.4 nm, whereas the modified PMMA film containing ZnO NPs had the lowest roughness factor (41.3 nm). The incorporation of metal oxide nanoparticles improved the roughness (Rq) of PMMA in this work compared to the films modified by the organometallic moiety incorporated into the polymer’s backbone (115.8–64.9 nm).10
|
Figure 1 FTIR spectra of blank PMMA film prior to and after irradiation. |
|
Figure 2 IOH values of PMMA films through the irradiation process. |
|
Figure 3 The percentage weight loss of PMMA films through irradiation. |
|
Figure 4 SEM images of irradiated PMMA film with 100 and 50 µm of magnification power: (a) Modified PMMA; (b) PMMA/NiO NPs; (c) PMMA/TiO2 NPs; (d) PMMA/MgO NPs; (e) PMMA/ZnO NPs films. |
|
Figure 5 AFM images of irradiated PMMA film: (a) chemically modified PMMA; (b) PMMA/NiO NPs; (c) PMMA/TiO2 NPs; (d) PMMA/MgO NPs; (e) PMMA/ZnO NPs films. |
|
Table 1 Roughness Factor (Rq; nm) of Modified PMMA Materials After 300 h of Irradiation |
The investigation demonstrates that the introduction of metal oxide nanoparticles onto the surface of poly(methyl methacrylate) chains can effectively increase their stability against UV irradiation. The nanoparticles investigated were nickel oxide, titanium dioxide, magnesium oxide, and zinc oxide. Modifying poly(methyl methacrylate) allowed for a significant reduction in the harmful effects of ultraviolet irradiation at a wavelength of 365 nm, extending protection for up to 300 hours. The effectiveness of the films was evaluated using FTIR spectroscopy, weight loss measurements, AFM, and SEM. The results showed that surface modification reduced the harmful effects of ultraviolet irradiation. Of the metals explored, the poly(methyl methacrylate) film containing zinc oxide nanoparticles showed the highest protection against photodegradation. Conversely, the film with the highest degradation was observed for the unmodified poly(methyl methacrylate) film.
- 1. Kanda, M.; Nishi, Y. Effects of Water Absorption on Impact Value of Aluminum Dispersed Composite Nylon 6. Mater. Trans. 2009, 50, 177-181.
-

- 2. Blasco, E.; Sims, M. B.; Goldmann, A. S.; Sumerlin, B. S.; Barner-Kowollik, C. 50th Anniversary Perspective: Polymer Function-alization. Macromolecules 2017, 50, 5215-5252.
-

- 3. Zyoud, S. H.; Almoadi, A.; Alabdulaal, T. H.; Alqahtani, M. S.; Harraz, F. A.; Al-Assiri, M. S.; Yahia, I. S.; Zahran, H. Y.; Mo-hammed, M. I.; Abdel-Wahab, M. S. Structural, Optical, and Electrical Investigations of Nd2O3-doped PVA/PVP Polymeric Composites for Electronic and Optoelectronic Applications. Polymers 2023, 15, 1351.
-

- 4. Ji, B. C.; Lee, E. M.; Yeum, J. H. Effect of Modification of Silver Nanoparticles on the Preparation of Poly(methyl methacrylate)/silver Microspheres via Suspension Polymerization. Polym. Korea 2010, 34, 341-345.
-

- 5. Chen, Q.; Yang, C.Q.; Zhao, T. The Chemical Bonding and Fire Performance of the Nylon/cotton Blend Fabrics Treated with a Hydroxy-functional Organophosphorus Oligomer. Polym. Degrad. Stab. 2016, 128, 237-244.
-

- 6. Ibrahim, B. A.; Kadum, K. M. Influence of Polymer Blending on Mechanical and Thermal Properties. Mod. Appl. Sci. 2010, 4, 157-161.
-

- 7. Teodorescu, M.; Bercea, M.; Morariu, S. Biomaterials of PVA and PVP in Medical and Pharmaceutical Applications: Perspectives and Challenges. Biotechnol. Adv. 2018, 37, 109-131.
-

- 8. El-Hiti, G. A.; Ahmed, D. S.; Yousif, E.; Al-Khazrajy, O. S. A.; Abdallh, M.; Alanazi, S. A. Modifications of Polymers Through the Addition of Ultraviolet Absorbers to Reduce the Aging Effect of Accelerated and Natural Irradiation. Polymers 2022, 14, 20.
-

- 9. Al-Sarray, A. J. A.; Jasim, B. E.; Aboud, N. A.-A.; Moaen, F. J. Enhancing the Photostability of Poly(vinyl chloride) (PVC) Through the Incorporation of Cerium and Samarium Oxide. Polym. Korea 2024, 48, 188-194.
-

- 10. Sansul, S.; Yousif, E.; Ahmed, D. S.; El-Hiti, G. A.; Kariuki, B. M.; Hashim, H.; Ahmed, A. Pendant Modification of Poly(methyl methacrylate) to Enhance Its Stability Against Photoirradiation. Polymers 2023, 15, 2989.
-

- 11. Thomas, S.; Grohens, Y.; Jyotishkumar, P. Characterization of Polymer Blends: Miscibility, Morphology and Interfaces; John Wiley & Sons: Weinheim, 2015.
- 12. Xavier, S. F. Properties and Performance of Polymer Blends. In Polymer Blends Handbook; Utracki, L.A. eds, Springer: Dordrecht, 2023.
- 13. Bartczak, Z.; Galeski, A. Mechanical Properties of Polymer Blends. In Polymer Blends Handbook; Utracki, L.; Wilkie, C. Eds, Springer: Dordrecht, 2014.
-

- 14. Lu, H.; Li, X.; Lei, Q. Conjugated Conductive Polymer Materials and Its Applications: A Mini-review. Front. Chem. 2021, 9, 732132.
-

- 15. Mdluli, S. B.; Ramoroka, M. E.; Yussuf, S. T.; Modibane, K. D.; John-Denk, V. S.; Iwuoha, E. I. π-Conjugated Polymers and Their Application in Organic and Hybrid Organic-silicon Solar Cells. Polymers 2022, 14, 716.
-

- 16. Pelin, G.; Sonmez, M.; Pelin, C.-E. The Use of Additive Manufacturing Techniques in the Development of Polymeric Molds: A Review. Polymers 2024, 16, 1055.
-

- 17. Delogu, F.; Gorrasi, G.; Sorrentino, A. Fabrication of Polymer Nanocomposites via Ball Milling: Present Status and Future Perspectives. Prog. Mater. Sci. 2017, 86, 75-126.
-

- 18. Prakash, S.; Kumar, S. Experimental Investigations and Analytical Modeling of Multi-pass CO2 Laser Processing on PMMA. Precis. Eng. 2017, 49, 220-234.
-

- 19. Junior, W. S.; Emmler, T.; Abetz, C.; Handge, U. A.; Santos, J. F.; Amancio-Filho, S. T.; Abetz, V. Friction Spot Welding of PMMA with PMMA/silica and PMMA/silica-g-PMMA Nanocomposites Functionalized via ATRP. Polymer 2014, 55, 5146-5159.
-

- 20. Arraq, R. R.; Hadi, A. G.; Ahmed, D. S.; El-Hiti, G. A.; Kariuki, B. M.; Husain, A. A.; Bufaroosha, M.; Yousif, E. Enhancement of Photostabilization of Poly(vinyl chloride) in the Presence of tin–cephalexin Complexes. Polymers 2023, 15, 550.
-

- 21. Naoom, N.; Yousif, E.; Ahmed, D. S.; Kariuki, B. M.; El-Hiti, G. A. Synthesis of Methyldopa–tin Complexes and Their Applicability as Photostabilizers for the Protection of Polyvinyl Chloride Against Photolysis. Polymers 2022, 14, 4590.
-

- 22. Fadhil, M.; Yousif, E.; Ahmed, D. S.; Kariuki, B. M.; El-Hiti, G. A. Synthesis and Application of Levofloxacin–tin Complexes as New Photostabilizers for Polyvinyl Chloride. Polymers 2022, 14, 3720.
-

- 23. Fadhil, M.; Yousif, E.; Ahmed, D. S.; Mohammed, A.; Hashim, H.; Ahmed, A.; Kariuki, B. M.; El-Hiti, G. A. Synthesis of New Norfloxacin–tin Complexes to Mitigate the Effect of Ultraviolet-visible Irradiation in Polyvinyl Chloride Films. Polymers 2022, 14, 2812.
-

- 24. Emad, N.; El-Hiti, G. A.; Yousif, E.; Kariuki, B. M. Metal Oxide Nanoparticles Containing Clotrimazole to Suppress Photodegradation of Poly(Vinyl chloride) Thin Films. Polymers 2023, 15, 1632.
-

- 25. Al-Sarray, A. J. A.; Jasim, B. E.; Aboud, N. A. A.; Moaen, F. J. Enhancing the Photostability of Poly(vinyl chloride)(PVC) Through the Incorporation of Cerium and Samarium Oxide. Polym. Korea 2024, 48, 188-194.
-

- 26. Jun, M.; Seo, J.-Y. Study on Thin Film Encapsulation Coating for Long-term Stable Organic Solar Cells. Polym. Korea 2023, 47, 87-91.
-

- 27. Yousif, E.; El-Hiti, G. A.; Haddad, R.; Balakit, A. A. Photochemical Stability and Photostabilizing Efficiency of Poly(methyl methacrylate) Based on 2-(6-methoxynaphthalen-2-yl)propanoate Metal Ion Complexes. Polymers 2015, 7, 1005-1019.
-

- 28. Gaumet, S.; Gardette, J.-L. Photo-oxidation of Poly(vinyl chloride): Part 2—A Comparative Study of the Carbonylated Products in Photochemical and Thermal Oxidations. Polym. Degrad. Stab. 1991, 33, 17-34.
-

- 29. Pospíšil, J.; Nešpurek, S. Photostabilization of Coatings. Mechanisms and Performance. Prog. Polym. Sci. 2000, 25, 1261-1335.
-

- 30. Brown, L.; Koerner, T.; Horton, J. H.; Oleschuk, R. D. Fabrication and Characterization of Poly(methyl methacrylate) Microfluidic Devices Bonded Using Surface Modifications and Solvents. Lab Chip 2006, 6, 66-73.
-

- 31. Soghli, I.; Khalaji, A.D.; Grivani, G. Copper (II) and Vanadium (IV) Complexes of New Modified Poly(vinyl chloride) Schiff Base for Catalytic Studies in Knoevenagel Condensation. Inorg. Chem. Res. 2021, 5, 163-172.
-

- 32. Manouras, T.; Argitis, P. High Sensitivity Resists for EUV Lithography: a Review of Material Design Strategies and Performance Results. Nanomaterials 2020, 10, 1593.
-

- 33. Daraboina, N.; Madras G. Thermal and Photocatalytic Degradation of Poly(methyl methacrylate), Poly(butyl methacrylate), and Their Copolymers. Ind. Eng. Chem. Res. 2008, 47, 6828-6834.
-

- 34. Zhang, Y.; Yang, J.; Fan, F.; Qing, B.; Zhu, C.; Shi, Y.; Fan, J.; Deng, X. Effect of Divalent Metals on the UV-shielding Properties of MII/MgAl Layered Double Hydroxides. ACS Omega 2019, 4, 10151-10159.
-

- 35. Silva, T. F.; Soares, B. G.; Ferreira, S. C.; Livi, S. Silylated Montmorillonite as Nanofillers for Plasticized PVC Nanocomposites: Effect of the Plasticizer. Appl. Clay Sci. 2014, 99, 93-99.
-

- 36. Zhao, Y.; Dan, Y. Preparation and Characterization of a High Molecular Weight UV-stabilizer Based on a Derivative of 2,4-dihydroxybenzophenone and Its Application in Polymer Aterials. J. Appl. Polym. Sci. 2006, 102, 2203-2211.
-

- 37. Spence, J. C. H. High-resolution Electron Microscopy; Oxford University Press: New York, 2013.
- 38. Yu, X.; Arey, B.; Chatterjee, S.; Chun J. Improving in situ Liquid SEM Imaging of Particles. Surf. Interface Anal. 2019, 51, 1325-1331.
-

- 39. Cohen, S. H.; Bray, M. T.; Lightbody, M. L. Atomic Force Microscopy/Scanning Tunneling Microscopy; Springer: Boston, 1994.
-

- 40. Binnig, G., Quate, C. F.; Gerber, C. Atomic Force Microscope. Phys. Rev. Lett. 1986, 56, 930-933.
-

- 41. Abdellatif, A. A. H.; Alhathloul, S. S.; Aljohani, A. S. M.; Maswadeh, H.; Abdallah, E. M.; Musa, K. H.; El Hamd, M. A. Green Synthesis of sIlver Nanoparticles Incorporated Aromatherapies Utilized for Their Antioxidant and Antimicrobial Activities Against Some Clinical Bacterial Isolates. Bioinorg. Chem. Appl. 2022, 11, 2432758.
-

- 42. He, T. Spongelike Structure. In: Drioli, E., Giorno, L. (eds) Encyclopedia of Membranes. Springer, Berlin, Heidelberg, 2016.
- 43. Hashim, H.; El-Hiti, G. A.; Alotaibi, M. H.; Ahmed, D. S.; Yousif, E. Fabrication of Ordered Honeycomb Porous Poly(vinyl chloride) Thin Film Doped with a Schiff Base and Nickel (II) Chloride. Heliyon 2018, 4, e00743.
-

- 44. Liu, C.-X.; Lang, W.-Z.; Shi, B.-B.; Guo, Y. J. Fabrication of Ordered Honeycomb Porous Polyvinyl Chloride (PVC) Films by Breath Figures Method. Mater. Lett. 2013, 107, 53-55.
-

- 45. Zhang, A.; Bai, H.; Li, L. Breath Figure: a Nature-inspired Preparation Method for Ordered Porous Films. Chem. Rev. 2015, 115, 9801-9868.
-

- 46. Alotaibi, M. H.; El-Hiti, G. A.; Hashim, H.; Hameed, A. S.; Ahmed, D. S.; Yousif, E. SEM Analysis of the Tunable Honeycomb Structure of Irradiated Poly(vinyl chloride) Films Doped With Polyphosphate. Heliyon 2018, 4, e01013.
-

- 47. Fu, M.; Li, D.; Liu, H.; Ai, H.; Zhang, Y.; Zhang, L. Synergistic Effects of Zinc-mannitol Alkoxide with Calcium/zinc Stearates and With b-diketone on Thermal Stability of Rigid Poly(vinyl chloride). J. Polym. Res. 2016, 23, 13.
-

- 48. Abd Kati, F. The Effects of TiO2 Nanoparticles on Flexural Strength of Self-polymerized Resins: In vitro Study. J. Tech. 2022, 4, 45-49.
-

- 49. Abellán-Nebot, J. V.; Vila Pastor, C.; Siller, H. R. A Review of the Factors Influencing Surface Roughness in Machining and Their Impact on Sustainability. Sustainability 2024, 16, 1917.
-

- Polymer(Korea) 폴리머
- Frequency : Bimonthly(odd)
ISSN 2234-8077(Online)
Abbr. Polym. Korea - 2024 Impact Factor : 0.6
- Indexed in SCIE
 This Article
This Article
-
2025; 49(2): 197-204
Published online Mar 25, 2025
- 10.7317/pk.2025.49.2.197
- Received on Sep 20, 2024
- Revised on Nov 29, 2024
- Accepted on Dec 12, 2024
 Services
Services
- Full Text PDF
- Abstract
- ToC
- Acknowledgements
- Conflict of Interest
Introduction
Experimental
Results and Discussion
Conclusions
- References
Shared
 Correspondence to
Correspondence to
- Gamal A. El-Hiti
-
Department of Optometry, College of Applied Medical Sciences, King Saud University, Riyadh 11433, Saudi Arabia
- E-mail: gelhiti@ksu.edu.sa
- ORCID:
0000-0001-6675-3126








 Copyright(c) The Polymer Society of Korea. All right reserved.
Copyright(c) The Polymer Society of Korea. All right reserved.