- Clamshell-shaped Droplet-based Electricity Generator with a Water Droplet Confined in a Wedge Plate
Department of Chemical Engineering, Chung-Ang University, 84 Heukseok-ro, Dongjak-gu, Seoul 06974, Korea
- 고정된 물방울을 이용한 조개 모양의 물방울 기반 전기 발전기
중앙대학교 화학공학과
Reproduction, stored in a retrieval system, or transmitted in any form of any part of this publication is permitted only by written permission from the Polymer Society of Korea.
Many previous studies on droplet-based electricity generators (DEGs) and triboelectric nanogenerators (TENG) used falling water droplets. These devices featured surfaces that were exposed to the environment, rendering them vulnerable to contaminants such as dust. They were also characterized with low durability. In such devices, high currents could be achieved only when the water droplets reached the electrode when they were spread out the most. Thus, further improvements in DEGs and TENGs must be implemented. In this study, an energy harvesting system was fabricated using a simple clamshell-shaped DEG, hereafter referred to as CS-DEG, prepared from polytetrafluoroethylene (PTFE) and polydimethylsiloxane films. The CS-DEG designed in this study does not generate electricity using falling water droplets; instead, it generates electricity through the compression and relaxation of a water droplet fixed to its surface. The developed design effectively prevented the contamination of the PTFE surface, and the resulting device demonstrated feasibility as a highly durable energy harvesting system. Specifically, it could generate a voltage of up to 0.9 V and a current of up to 711 nA using a single water droplet. The proposed technology has potential for further development and application in various fields.
기존의 많은 연구에서 물방울 기반 전기 발생기(electricity generators, DEG) 및 마찰 전기 나노 발전기(triboelectric nanogenerators, TENG)는 주로 낙하하는 물방울을 이용하여 에너지를 생성하였다. 이러한 장치들은 작동 중 외부 환경에 노출되어 먼지 등 오염 물질에 취약하며, 내구성 또한 낮다는 단점이 있다. 또한, 높은 전류를 얻기 위해서는 물방울이 전극 표면에 최대한 넓게 퍼질 때만 효율적인 전력 생산이 가능하다는 한계가 존재한다. 이러한 문제점을 해결하기 위해 DEG 및 TENG 기술의 추가적인 개선이 필요하다. 본 연구에서는 폴리테트라플루오로에틸렌(polytetrafluoroethylene, PTFE)과 폴리디메틸실록산(polydimethylsiloxane, PDMS) 필름을 이용하여 조개 모양의 간단한 구조를 갖춘 DEG 시스템(CS-DEG)을 설계하였다. CS-DEG는 낙하하는 물방울 대신, 표면에 고정된 물방울의 압축과 이완에 의해 전기를 생성하는 방식으로 작동한다. 이 설계는 PTFE 표면의 오염을 효과적으로 방지하였으며, 이를 통해 장치는 높은 내구성을 지닌 에너지 하베스팅 시스템으로서의 가능성을 입증하였다. 본 연구에서 제안된 시스템은 단일 물방울을 사용하여 최대 0.9 V의 전압과 711 nA의 전류를 생성할 수 있음을 확인하였다. 이러한 기술은 다양한 환경에서 안정적으로 에너지를 생산하는 효율적인 에너지 하베스팅 시스템으로 활용 가능성이 높다고 판단된다.
A novel clamshell-shaped droplet-based electricity generator (CS-DEG) was developed to harvest energy from water droplets through contact electrification and electrostatic induction. Unlike conventional triboelectric nanogenerators (TENGs), which are less effective without rainfall, the CS-DEG compresses and decompresses water droplets, generating electricity. Its hydrophobic design prevents contamination and ensures stable performance in diverse environments, making it a promising solution for sustainable energy generation.
Keywords: droplet-based electricity generator, water droplet, polytetrafluoroethylene film, energy harvesting.
This research was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. 2022R1A2C1005739), a Korea Institute of Advancement of Technology (KIAT) grant funded by the Korean government (MOTIE) (No. P0012453, HRD Program for Industrial Innovation), and a Chung-Ang University research grant awarded in 2024.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Advancements in society have led to increased demands for electricity generation, which requires a corresponding increase in the consumption of fossil fuels, leading to energy shortages, environmental degradation, and increased greenhouse gas emissions. Renewable energy is crucial in addressing these issues. Hydropower is a renewable energy source that utilizes the kinetic energy of water to generate electricity. Water is a clean and inexhaustible resource. However, much of the water energy available on Earth remains untapped. In addition, current hydropower systems are limited by high costs and low efficiency, and other forms of water energy, such as waves, tides, and river currents, have not been fully developed. Overcoming these challenges to improve the efficiency of hydropower utilization could significantly contribute to solving energy shortages and environmental issues.1-6
Droplet-based electricity generators (DEGs) are an innovative approach for harvesting energy from water droplets and converting the kinetic energy from falling or moving water into electrical energy. These devices have a simple structure and can be manufactured using inexpensive materials. They require no fossil fuels and do not generate harmful emissions. DEGs operate via the triboelectric effect, which involves the generation of electric charges through friction.3,7-13 When a water droplet strikes a surface, charge separation is induced if the surface is made of or coated with materials that have different affinities for electrons. The interaction between the droplet and surface induces charge separation owing to the triboelectric effect. The induced charges are collected and transferred to an electrode, generating a small current. The electricity generated can then be stored in a capacitor or battery for later use.14-25
Polymers such as polytetrafluoroethylene (PTFE) and polydimethylsiloxane (PDMS) are characterized with high triboelectric performance. Triboelectric nanogenerators (TENGs), which produce electricity using the mechanical movement of rain or falling water droplets, represent an innovative technology that is attracting attention for its potential use in various fields.26,27 However, the long-term durability of materials used in TENGs under varying environmental conditions requires improvement. Thus, further research on TENGs to improve their efficiency, enhance their durability, and promote their applications is necessary.
In this study, we developed a novel clamshell-shaped DEG, hereafter referred to as CS-DEG, that can harvest energy through the mechanical motion of water droplets. Similar to other TENG-based DEGs, our CS-DEG produces electrical energy through contact electrification and electrostatic induction. Conventional TENGs based on solid–liquid interfaces are ineffective in environments without rainfall or droplet impact. Unlike these devices, our CS-DEG operates based on the change in contact area of fixed water droplets following their compression. The CS-DEG is designed such that the device is not directly exposed to the external environment, thereby addressing the issue of contamination, which affects traditional TENG-based DEGs. The hydrophobic polymer layer of the CS-DEG ensures that water droplets do not easily adhere to the device surface, allowing for the smooth compression and decompression of the droplets. Charges are induced when a water droplet makes contact with a hydrophobic surface. As the droplets are compressed and decompressed, the movement of these charges generates electricity. The CS-DEG can stably harvest energy regardless of the environmental conditions, overcoming the limitations of traditional TENG-based DEGs. Thus, it is potentially useful for sustainably generating electricity in various environments.
Fabrication of a PTFE Film on an ITO Glass. An ITO glass substrate (40 × 40 × 0.7 mm, OMNISCIENCE, Korea) was washed with water and acetone and dried with nitrogen gas. Next, 3 mL of PTFE (Sigma-Aldrich, USA) was dropped onto the dried ITO glass using a pipette and then deposited through spin-coating at rates of 500, 750, 1000, and 1250 rpm. The coated substrate was then heated in a digital muffle furnace (DAIHAN SCIENTIFIC, Korea) at 350 ℃ for 1 h. Finally, the PTFE-coated ITO glass was cooled to room temperature. Field-emission scanning electron microscopy (Carl Zeiss, Germany) was used to obtain images of the PTFE film surfaces.
Fabrication of a PDMS Layer on a Glass Substrate. A glass substrate (76 mm × 52 mm × 1.5 mm, Paul Marienfeld GmbH & Co., Germany) was plasma-treated in a plasma cleaner (PDC-32gG, Harrick Plasma, USA) for 1.5 min. Ten grams of PDMS prepolymer (Sylgard 184 A, Dow Corning, USA) was mixed with 2 g of a curing agent (Sylgard 184 B, Dow Corning) at a 5:1 ratio, and the resulting compound was mixed with toluene at a mass ratio of 1:1. The mixed solution was deposited on the clean glass substrate by spin-coating at 1000 rpm. PDMS was cured in a ThermoStableTM OV vacuum oven (DAIHAN SCIENTIFIC, Korea) at 100 ℃ for 2 h. Finally, the PDMS-coated glass was cooled to room temperature.
Fabrication of the CS-DEG. Copper (Cu) tape was attached to a corner of the PTFE-coated ITO glass, and Teflon tape was wrapped around the edges of the glass. The CS-DEG was then covered by the PTFE-coated glass. The electrical energy produced by the CS-DEG was measured using a digital multimeter (34401A, Keysight, Korea) with a preamplifier (SR570, Stanford Research Systems, USA).
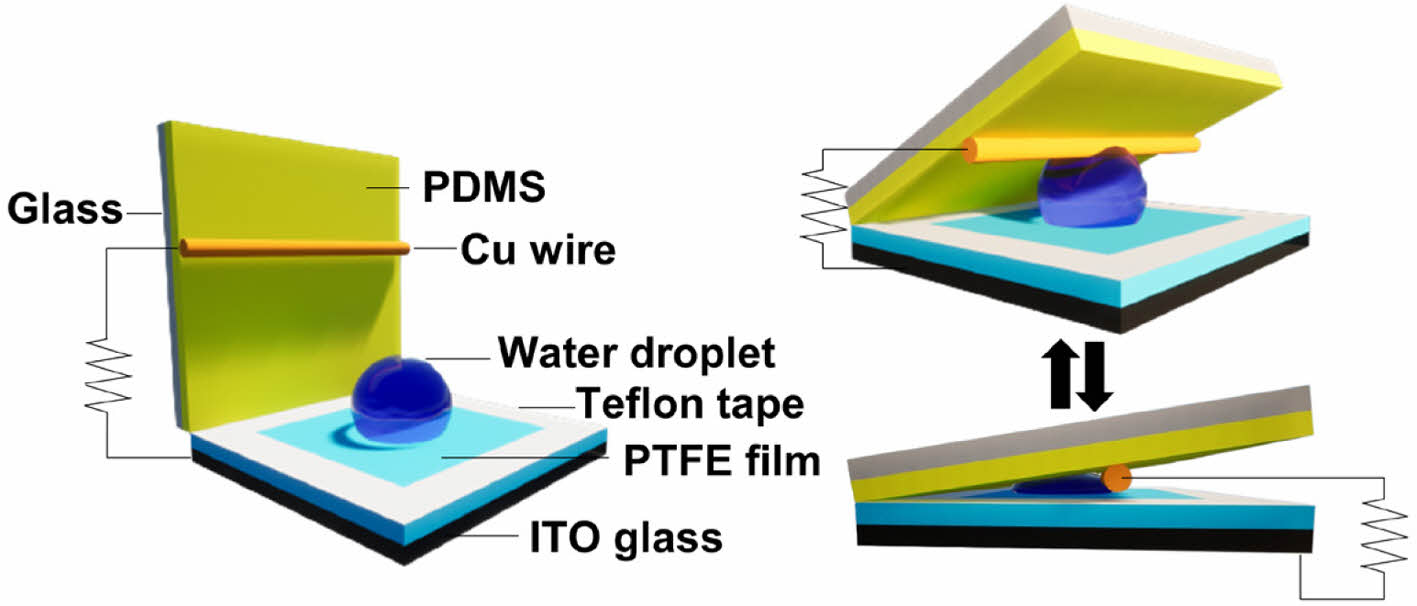
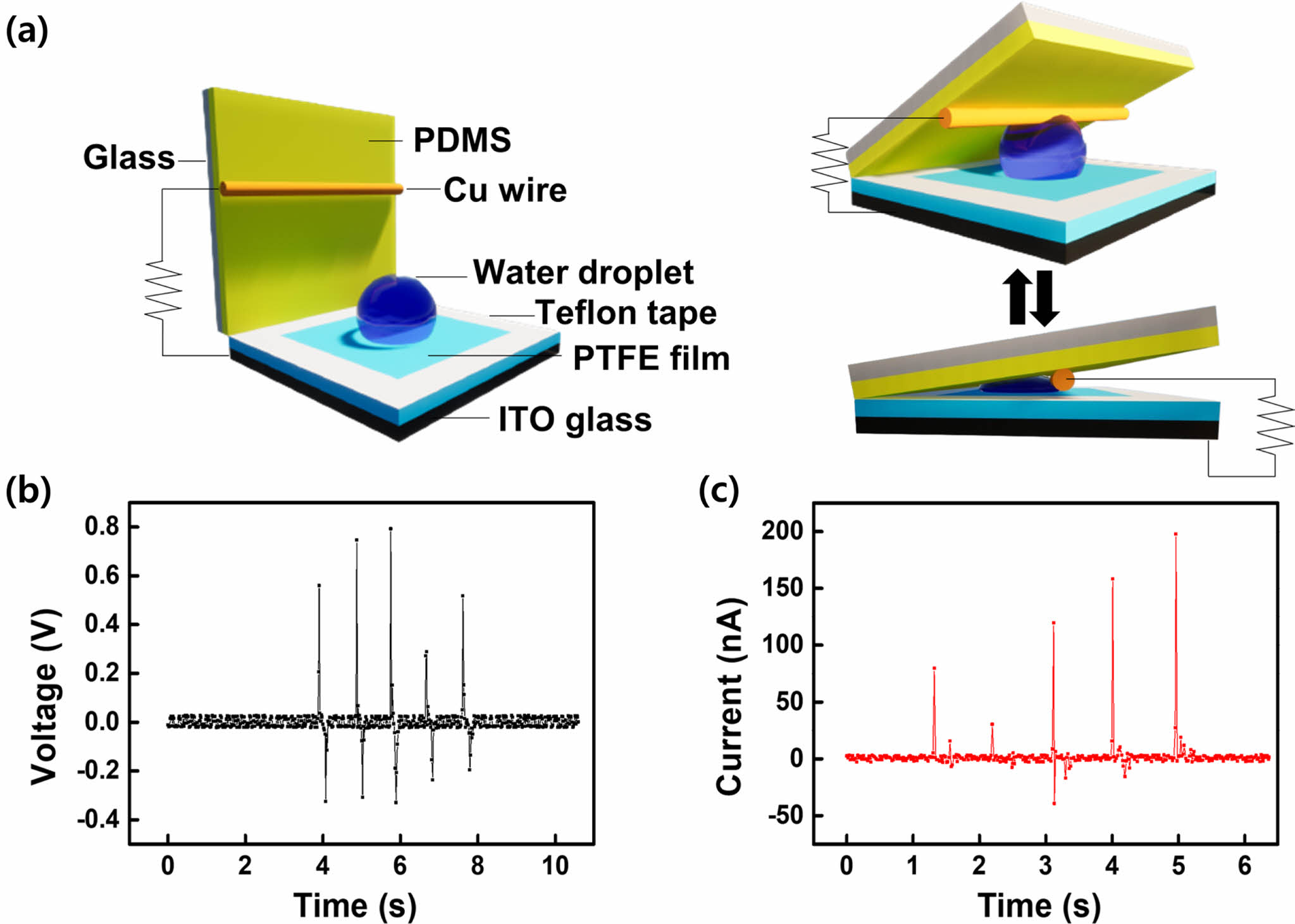
Figure 1(a) illustrates the design and functions of the CS-DEG. PTFE has a high dielectric constant and can conduct electricity when exposed to moisture. Furthermore, it is superhydrophobic, effectively preventing water droplets from sliding owing to friction with PDMS. The device features two overlapping polymer-covered glass layers such that it can change the contact area of water droplets during compression and relaxation and harvest electrical energy via contact electrification and electrostatic induction.28 A voltage and current are generated by the contact between a water droplet fixed to the PTFE-coated ITO glass and a copper wire attached to the PDMS-coated glass. PDMS, which has a lower dielectric constant than PTFE, reduces the voltage difference between the generated charges. Teflon tape was used to prevent contact between the PDMS and PTFE films. In the case of TENGs based on solid–liquid interfaces, electricity is generated through contact electrification, which occurs when water droplets fall. However, in environments where insufficient rain or water is available, the efficiency of solid–liquid-based TENGs decreases significantly, and they may not function effectively. Hence, solid–liquid-based TENGs are primarily used in environments with a continuous supply of water. Owing to the possibility of contamination from direct exposure to the environment, devices based on solid–liquid interfaces can face stability challenges in areas with frequent rain or a constant water supply. Unlike these devices, our CS-DEG employs a technique in which a small amount of kinetic energy is applied to generate electrical energy, instead of actively depending on falling water or rainfall. A potential for continuous electricity generation can be induced by adjusting the contact area of the fixed water droplet. Furthermore, the device minimizes the exposure of the active surface to the external environment and provides resistance to contamination. The movement of the PDMS-coated glass can be carefully controlled by an attached weight. The electrical energy generated by compressing and releasing the PDMS-coated glass substrate can be measured based on its weight. The PDMS-coated glass was consistently moved by hand pressure on the extended part. A voltage and current are generated when the PDMS-coated glass is pressed by a weight, as shown in Figures 1(b) and (c). The voltage and current were generated by the change in form of a 20 μL water droplet between the PDMS-coated glass and PTFE-coated ITO glass under a 1500 N force. Positive and negative charges were generated during compression and release, respectively. The device generated a maximum voltage of 0.8 V and a current of 200 nA using a 20 µL water droplet.
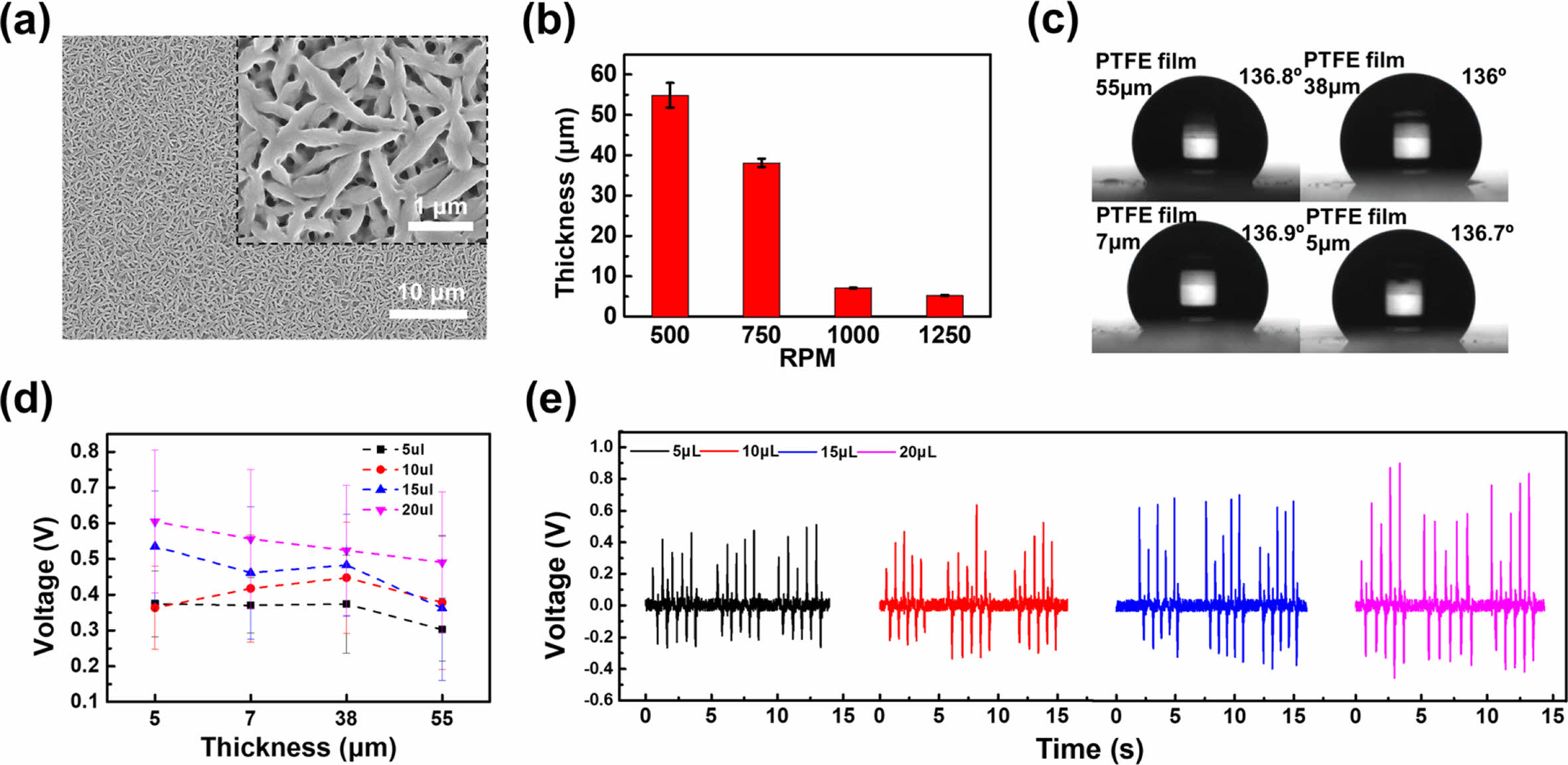
Figure 2 shows the electrical energy generated by the device according to the thickness of the superhydrophobic PTFE film. The PTFE solution was deposited onto the ITO glass using the spin-coating method. An image of the PTFE film spin-coated onto the ITO glass at a rate of 1250 rpm is shown in Figure 2(a). As the most electronegative material in the triboelectric series, a porous PTFE layer with an average pore size of 200 nm was created on the ITO glass. The PTFE film was formed by heating the coated ITO glass in a furnace and cooling to room temperature. The thicknesses of PTFE layers deposited by spin-coating at rates of 500, 750, 1000, and 1250 rpm are shown in Figure 2(b). We measured the contact angles of 20 μL water droplets on PTFE substrates with thicknesses of 55, 38, 7, and 5 µm and obtained values of 136.7°, 136.7°, 136.9°, and 136.8°, respectively (Figure 2(c)). Electrical energy variations were analyzed based on the contact area of water droplets of different sizes as a function of PTFE layer thickness, as shown in Figure 2(d). The average voltage was then determined in relation to the PTFE thickness. A 20 μL water droplet generated average voltages of 0.6 V with a 5 μm-thick PTFE layer and 0.52 V with a 55 μm-thick PTFE layer. The voltage decreased as the PTFE thickness increased. However, the average voltage generated with a 10 μL water droplet and PTFE layers of different thicknesses showed no remarkable differences, measuring 0.37 V for a 5 μm-thick PTFE layer and 0.4 V for a 55 μm-thick PTFE layer. These results indicate that the PTFE thickness does not significantly affect the voltage generated over multiple contacts. However, the maximum voltage increased with the volume of the water droplet, indicating that more electrical energy is generated by larger droplets because they have larger contact surfaces. Smaller droplets tend to have a higher charge density because their surface area is larger than their volume; by contrast, larger droplets have a lower charge density.29 Therefore, the size of a droplet can significantly influence its electrical properties. The voltage generated as a function of the volume of a water droplet was measured by compressing and releasing the droplets five times and repeating this process three times with a 55 μm-thick PTFE film (Figure 2(e)). The change in electrical energy depending on the droplet volume increased as the droplet volume increased, resulting in higher voltages and currents regardless of the thickness of the PTFE film. Voltages of 0.3 and 0.48 V were generated by 5 and 20 μL water droplets, respectively, with 55 μm-thick PTFE film.
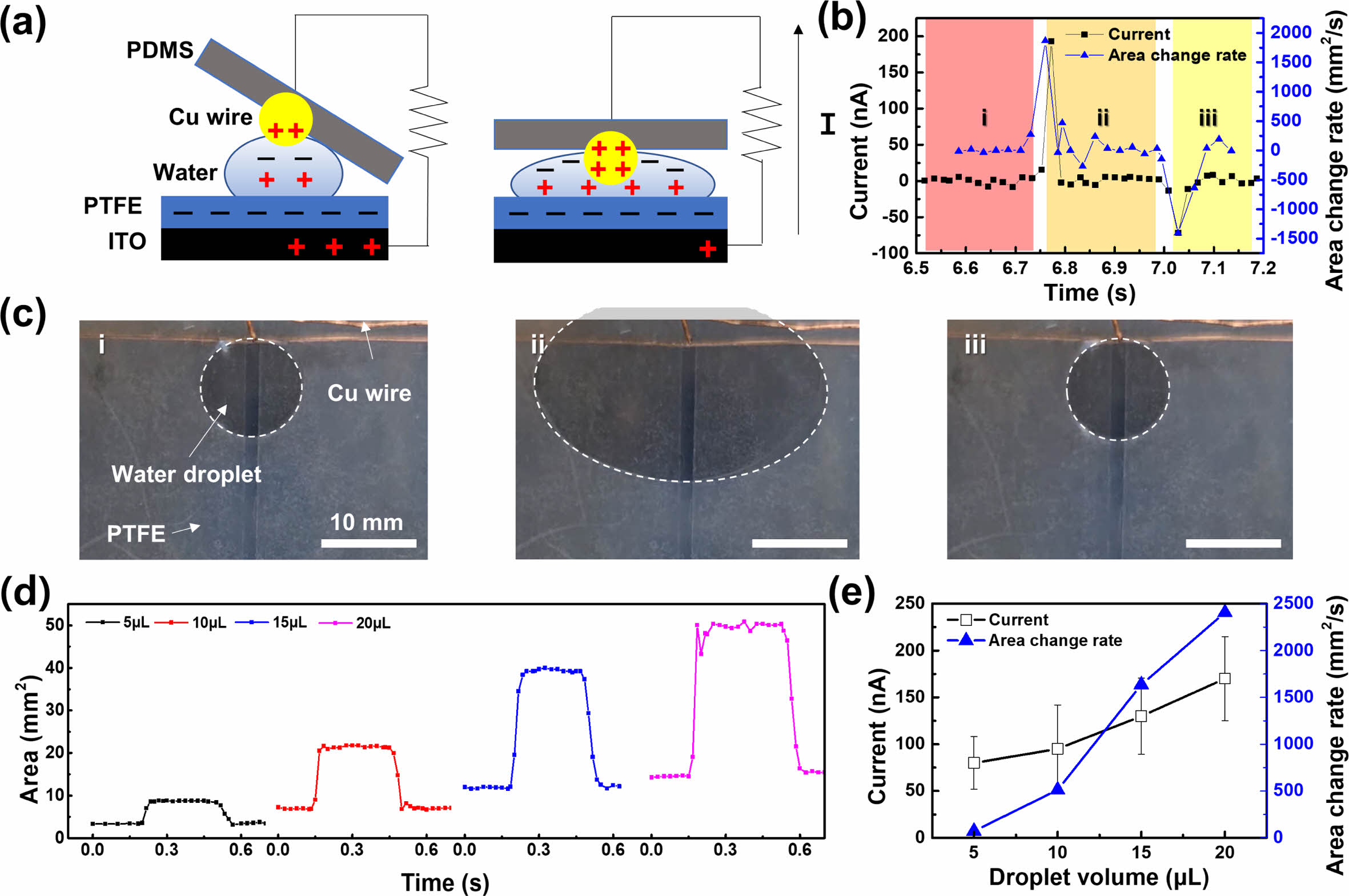
Figure 3(a) shows a schematic of the working principle of the CS-DEG. The device employed a technology similar to that in TENGs. A TENG generates electricity through friction and contact between different materials. When a water droplet comes into contact with the PTFE film, electrons are transferred from the droplet to PTFE, leaving the water droplet with a net negative charge. However, a water droplet can exhibit an asymmetrical charge distribution. Generally, the bottom part of the droplet in direct contact with the PTFE film becomes positively charged, whereas the top part of the droplet remains negatively charged, because of charge redistribution on the droplet surface after contact electrification. When a Cu wire comes into contact with the tip of a water droplet, electrical energy is generated on the PTFE film deposited on the ITO glass. Charge transfer occurs between the Cu wire and negatively charged upper portion of the water droplet. The contact area of the water droplet expands as it is forced onto the PDMS glass. Thus, a more negative charge reaches the PTFE surface and positive charges are generated on the ITO surface. These charges then flow to the Cu wire through the circuit, and a current is generated. When the Cu wire is released, the contact area between the PTFE and water droplet decreases, resulting in a decrease in charge. An opposing current flow forms as a result of the positive charge on the Cu wire moving back to the ITO substrate. Figures 3(b) and (c) show the currents generated according to the change in contact area between a water droplet and 55 μm-thick PTFE film. The area change of a water droplet with a diameter of 10 mm under a force of 1500 N was 1867 mm²/s, resulting in a current of 193 nA. When relaxed, the area change rate was -1408 mm²/s, with a corresponding current of -71 nA. Figure 3(d) shows the change in contact area of water droplets as a function of their volume under the same conditions. The application of the same force resulted in an increase in contact area, with droplets measuring 5, 10, 15, and 20 μL in volume having contact areas of approximately 28, 154, 572, and 962 mm2, respectively; the corresponding currents were 80, 95, 130, and 170 nA, respectively. Under the same applied force, the contact area changed proportionally to the size of the water droplet. Larger droplets have larger surface areas; therefore, applying the same force can cause more substantial changes in their contact area owing to the proportional relationship between the volume and surface area of a droplet. Therefore, the change in contact area of a water droplet is proportional to its size, and the resulting current increases proportionally, as shown in Figure 3(e).
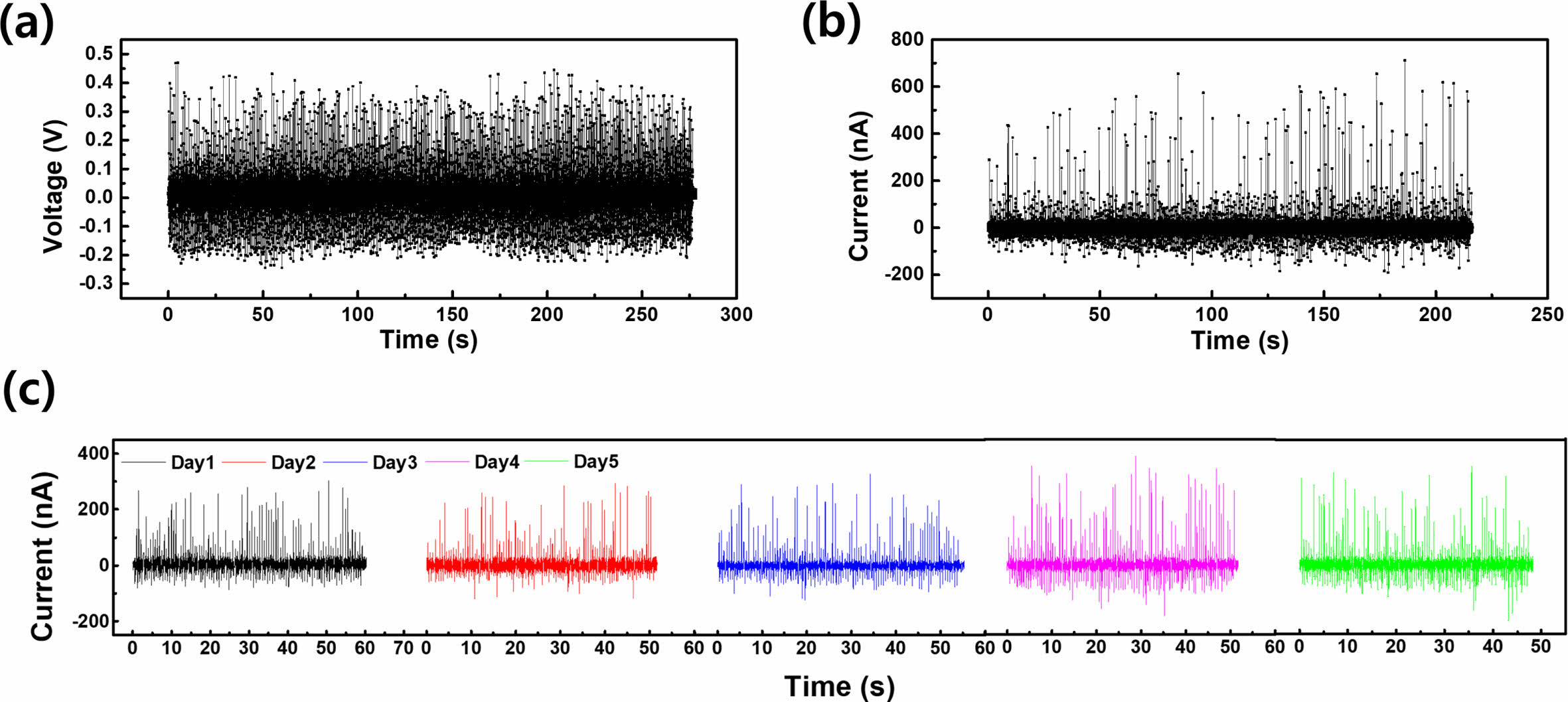
We evaluated the stability of our CS-DEG by performing 500 press–release cycles on a 20 μL water droplet. As shown in Figure 4(a), the voltage generated consistently remained between 0.2 and 0.4 V. Similarly, as shown in Figure 4(b), a current of up to 711 nA was generated, and the current was maintained above 200 nA. The performance of the CS-DEG was evaluated over a five-day period while cycling 100 times daily. Figure 4(c) shows that the PDMS-coated glass layer of the CS-DEG protected it from external contamination. Throughout the 5 days of cycling, a current of above 200 nA was consistently generated.
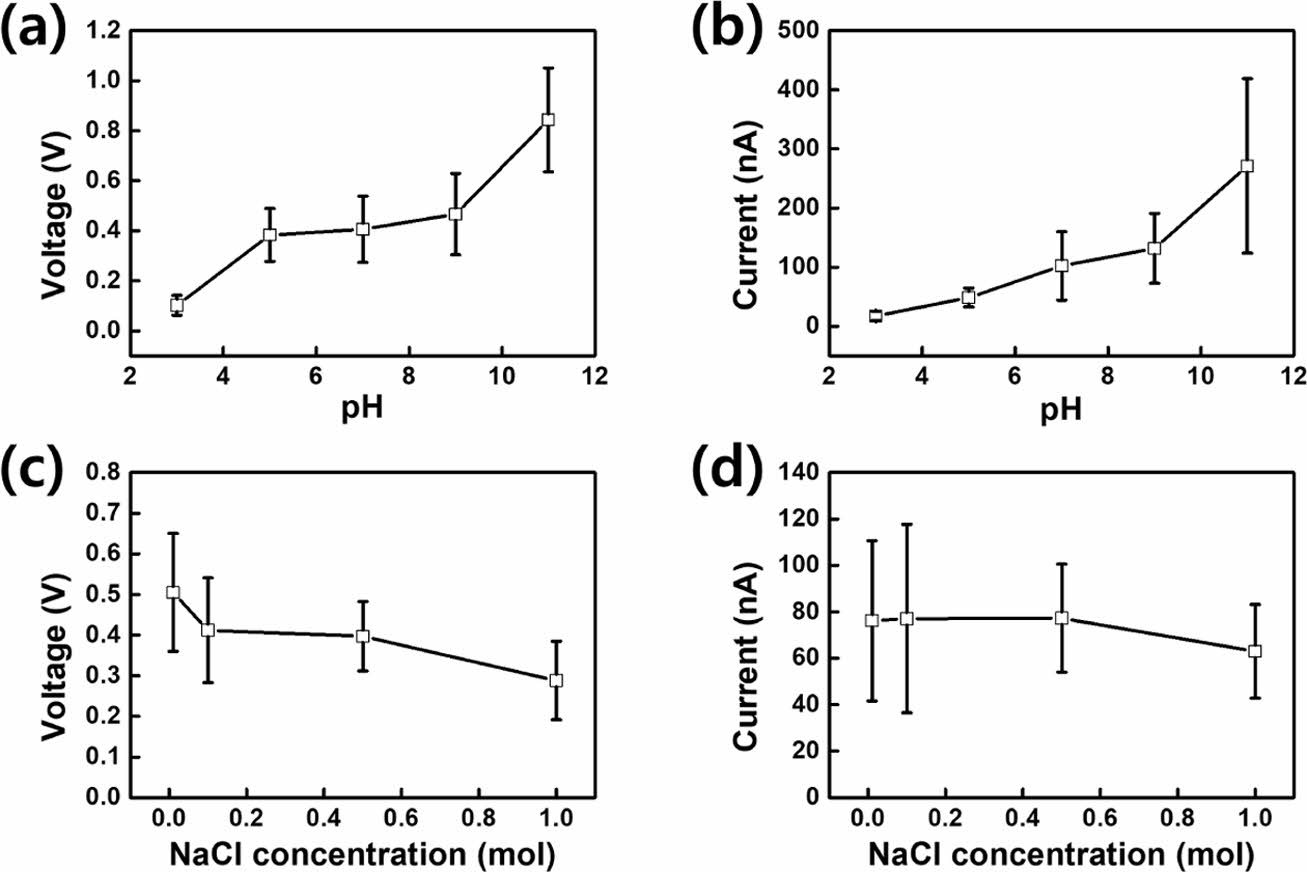
The electrical energy generated by a DEG can be influenced by the solution pH. Thus, the solution pH must be considered when evaluating and enhancing the performance of the CS-DEG. The increase in current generation at higher pH values is primarily due to the increased electrical conductivity of the solution.
Figures 5(a) and (b) indicate that as the pH concentration increases, the amount of electrical energy generated increases. At a higher pH, ions can move freely, facilitating more active charge transfer and leading to an increase the current generated by the DEG.30 When a water droplet makes contact with the PTFE surface, the PTFE surface becomes negatively charged. At this point, the Na+ and Cl− ions in the NaCl solution move because of the electrical interaction between the water droplet and PTFE surface, partially or fully neutralizing the negative charge on the latter. As the ion concentration increases with increasing NaCl concentration, more of these interactions occur, leading to a greater degree of neutralization of the negative charge on the PTFE surface. This neutralization of the negative charge on the PTFE surface can reduce the potential difference between the PTFE surface and Cu electrode or ITO layer, leading to a decrease in current.31 As the concentration of the electrolyte increases, these interactions increase, resulting in a decrease in electrical energy production. As shown in Figures 5(c) and (d), as the concentration of the NaCl solution increases, the electrical energy produced by the DEG decreases. These results support the working mechanism of the CS-DEG and demonstrate its applicability to different liquids. The findings in this study should be carefully considered when designing solid–liquid TENGs for both energy harvesting and sensing devices.
|
Figure 1 (a) Schematic diagram of the design and working principle of the CS-DEG; (b) voltage; (c) current generated by a 5 μm-thick PTFE film and 20 µL water droplet. |
|
Figure 2 Characterization of the PTFE-coated ITO glass: (a) FE-SEM images of the surface of a 5 μm-thick PTFE film; (b) PTFE thickness as a function of spin-coating speed; (c) contact angle of a 20 µL water droplet as a function of PTFE thickness; (d) voltage generated as a function of PTFE thickness for water droplets of different volumes; (e) voltage generated as a function of droplet size on the surface of a 55 μm-thick PTFE film. |
|
Figure 3 Analysis of the mechanism of the CS-DEG: (a) schematic of the working mechanism of the CS-DEG; (b) measurements of the change in contact area of the water droplets and the resulting current; (c) images of the change in contact area of a water droplet; (d) changes in contact area as a function of water droplet size; (e) correlation between the change in contact area and resulting current as a function of water droplet volume. |
|
Figure 4 Stability evaluation of the CS-DEG: (a) voltage; (b) current generated with a 20 µL water droplet over 500 cycles; (c) current variation over 5 days at 100 cycles per day |
|
Figure 5 Evaluation of the electricity generation performance of the CS-DEG as function of the solution pH and electrolyte concentration of water droplets. Measured (a) voltage; (b) current as a function of pH. Measured (c) voltage; (d) current as a function of NaCl concentration. |
We designed a CS-DEG based on the compression and relaxation of water droplets between hydrophobic (PDMS) and superhydrophobic (PTFE) surfaces with varying dielectric constants to harvest energy from the contact area of a water droplet. Various PTFE thicknesses and water droplet sizes were investigated. Although the PTFE thickness did not significantly affect the electrical energy generated by the DEG, larger water droplets led to increased energy production. For instance, a 20 μL water droplet compressed under a force of 1500 N yielded a contact area of 962 mm2 and generated a current of 170 nA. The stability of the CS-DEG was evaluated over 500 press–release cycles, and currents of up to 711 nA were consistently produced. Moreover, the generated currents were generally maintained above 200 nA. Variations in electrical energy production owing to changes in solution pH and electrolyte concentration were analyzed to assess the stability and characteristics of the device. The PDMS-coated layer prevented contamination from external sources and maintained the contact area of the substrate to maximize energy production. The CS-DEG operates by utilizing kinetic energy to generate electrical energy, rather than relying on falling water or rainfall. Thus, the device is an innovative electricity generator that can overcome the limitations of previous devices by harvesting energy from fixed water droplets and addressing contamination issues using a hydrophobic polymer layer. This technology has the potential for further development and application in various fields.
- 1. Zheng, Y.; Li, J.; Xu, T.; Cui, H.; Li, X. Triboelectric Nanogenerator for Droplet Energy Harvesting Based on Hydrophobic Composites. Materials (Basel). 2023, 16, 5439-5449.
-

- 2. Li, W.; Zhang, B.; Yuan, Z.; Shen, P.; Yu, X.; Wang, Z. L.; Cheng, T. IEEE Trans. Enhancing Energy Storage and Utilization of Triboelectric Nanogenerator by a Synchronized Charge Accumulation Circuit. Power Electron. 2023, 38, 9294-9301.
-

- 3. Li, X.; Ning, X.; Li, L.; Wang, X.; Li, B.; Li, J.; Yin, J.; Guo, W. Performance and Power Management of Droplets-based Electricity Generators. Nano Energy 2022, 92, 106705-106712.
-

- 4. Xu, W.; Zhou, X.; Hao, C.; Zheng, H.; Liu, Y.; Yan, X.; Yang, Z.; Leung, M.; Zeng, X. C.; Xu, R. X.; SLIPS-TENG, Z. Robust Triboelectric Nanogenerator with Optical and Charge Transparency Using a Slippery Interface. Natl. Sci. Rev. 2019, 6, 540-550.
-

- 5. Rehman, S.; Al-Hadhrami, L. M.; Alam, M. M. Pumped Hydro Energy Storage System: A Technological Review. Renew. Sustain. Energy Rev. 2015, 44, 586-598.
-

- 6. Wang, Y.; Gao, S.; Xu, W.; Wang, Z. Nanogenerators with Superwetting Surfaces for Harvesting Water/Liquid Energy. Adv. Funct. Mater. 2020, 30, 1908252-1908266.
-

- 7. Xu, W.; Zheng, H.; Liu, Y.; Zhou, X.; Zhang, C.; Song, Y.; Deng, X.; Leung, M.; Yang, Z.; Xu, R. X.; Wang, Z. L.; Zeng, X. C.; Wang, Z. A Droplet-based Electricity Generator with High Instantaneous Power Density. Nature 2020, 578, 392-407.
-

- 8. Wang, X.; Fang, S.; Tan, J.; Hu, T.; Chu, W.; Yin, J.; Zhou, J.; Guo, W. Dynamics for Droplet-based Electricity Generators. Nano Energy 2021, 80, 105558-105563.
-

- 9. Zhang, N.; Gu, H.; Lu, K.; Ye, S.; Xu, W.; Zheng, H.; Song, Y.; Liu, C.; Jiao, J.; Wang, Z.; Zhou, X. A Universal Single Electrode Droplet-based Electricity Generator (SE-DEG) for Water Kinetic Energy Harvesting. Nano Energy 2021, 82, 105735-105743.
-

- 10. J. Dong, C. Xu, L. Zhu, X. Zhao, H. Zhou, H. Liu, G. Xu, G. Wang, G. Zhou, Q. Zeng, Q. Song, A high voltage direct current droplet-based electricity generator inspired by thunderbolts. Nano Energy 2021, 90, 106567-106576.
-

- 11. Li, Z.; Yang, D.; Zhang, Z.; Lin, S.; Cao, B.; Wang, L.; Wang, Z. L.; Yin, F. A Droplet-based Electricity Generator for Large-scale Raindrop Energy Harvesting. Nano Energy 2022, 100, 107443-107449.
-

- 12. Wang, L.; Song, Y.; Xu, W.; Li, W.; Jin, Y.; Gao, S.; Yang, S.; Wu, C.; Wang, S.; Wang, Z. Harvesting Energy from High-frequency Impinging Water Droplets by a Droplet-based Electricity Generator. EcoMat 2021, 3, 12116-12124.
-

- 13. Liu, C.; Sun, J.; Zhuang, Y.; Wei, J.; Li, J.; Dong, L.; Yan, D.; Hu, A.: Zhou, X.; Wang, Z. Self-propelled Droplet-based Electricity Generation. Nanoscale 2018, 10, 23164-23169.
-

- 14. Choi, D.; Lee, S.; Park, S. M.; Cho, H.; Hwang, W.; Kim, D. S. Energy Harvesting Model of Moving Water Inside a Tubular System and its Application of a Stick-type Compact Triboelectric Nanogenerator. Nano Res. 2015, 8, 2481-2491.
-

- 15. Nanogenerator, T.; Cheng, G.; Lin, Z.; Du, Z.; Wang, Z. L. Simultaneously Harvesting Electrostatic and Mechanical Energies from Flowing Water by a Hybridized Triboelectric Nanogenerator. ACS Nano 2014, 8, 1932-1939.
-

- 16. Yao, Y.; Jiang, T.; Zhang, L.; Chen, X.; Gao, Z.; Wang, Z. L. Charging System Optimization of Triboelectric Nanogenerator for Water Wave Energy Harvesting and Storage. ACS Appl. Mater. Interfaces 2016, 8, 21398-21406.
-

- 17. Liu, Y.; Liu, W.; Wang, Z.; He, W.; Tang, Q.; Xi, Y.; Wang, X.; Guo, H.; Hu, C. Quantifying Contact Status and the Air-breakdown Model of Charge-excitation Triboelectric Nanogenerators to Maximize Charge Density. Nat. Commun. 2020, 11, 1599-1606.
-

- 18. Y. Su, Y. Yang, X. Zhong, H. Zhang, Z. Wu, Y. Jiang, Z. L. Wang, Fully Enclosed Cylindrical Single-Electrode-Based Triboelectric Nanogenerator. ACS Appl. Mater. Interfaces 2014, 6, 553-559.
-

- 19. Lin, Z. H.; Cheng, G.; Lin, L.; Lee, S.; Wang, Z. L. Water-Solid Surface Contact Electrification and its Use for Harvesting Liquid Wave Energy. Angew. Chemie - Int. Ed. 2013, 52, 12545-12550.
-

- 20. Liang, Q.; Yan, X.; Gu, Y.; Zhang, K.; Liang, M.; Lu, S.; Zheng, X.; Zhang, Y. Highly Transparent Triboelectric Nanogenerator for Harvesting Water-related Energy Reinforced by Antireflection Coating. Sci. Rep. 2015, 5, 9080-9086.
-

- 21. Kim, S.; Gupta, M. K.; Lee, K. Y.; Sohn, A.; Kim, T. Y.; Shin, K. S.; Kim, D.; Kim, S. K.; Lee, K. H.; Shin, H. J.; Kim, D. W.; Kim, S. W. Transparent Flexible Graphene Triboelectric Nanogenerators. Adv. Mater. 2014, 26, 3918-3925.
-

- 22. Chatterjee, S.; Burman, S. R.; Khan, I.; Saha, S.; Choi, D.; Lee, S.; Lin, Z. H. Recent Advancements in Solid–liquid Triboelectric Nanogenerators for Energy Harvesting and Self-powered Applications. Nanoscale 2020, 12, 17663-17697.
-

- 23. Quan, T.; Wu, Y.; Yang, Y. Hybrid Electromagnetic–triboelectric Nanogenerator for Harvesting Vibration Energy. Nano Res. 2015, 8, 3272-3280.
-

- 24. Khan, U.; Kim, S. W. Triboelectric Nanogenerators for Blue Energy Harvesting. ACS Nano 2016, 10, 6429-6432.
-

- 25. Wang, X.; Wen, Z.; Guo, H.; Wu, C.; He, X.; Lin, L.; Cao, X.; Wang, Z. L. Fully Packaged Blue Energy Harvester by Hybridizing a Rolling Triboelectric Nanogenerator and an Electromagnetic Generator. ACS Nano 2016, 10, 11369-11376.
-

- 26. Yoo, D.; Kim, S. J.; Joung, Y.; Jang, S.; Choi, D.; Kim, D. S. Lotus Leaf-inspired Droplet-based Electricity Generator with Low-adhesive Superhydrophobicity for a Wide Operational Droplet Volume Range and Boosted Electricity Output. Nano Energy 2022, 99, 107361-107372.
-

- 27. Lin, Z. H.; Cheng, G.; Lee, S.; Pradel, K. C.; Wang, Z. L. Harvesting Water Drop Energy by a Sequential Contact-Electrifi cation and Electrostatic-Induction Process. Adv. Mater. 2014, 26, 4690-4696.
-

- 28. Banpurkar, A. G.; Sawane, Y.; Wadhai, S. M.; Murade, C. U.; Siretanu, I.; Van Den Ende, D.; Mugele, F. Spontaneous Electrification of Fluoropolymer–water Interfaces Probed by Electrowetting. Faraday Discuss. 2017, 199, 29-47.
-

- 29. Yang, L.; Wang, Y.; Guo, Y.; Zhang, W.; Zhao, Z. Robust Working Mechanism of Water Droplet-Driven Triboelectric Nanogenerator: Triboelectric Output versus Dynamic Motion of Water Droplet. Adv. Mater. Interfaces 2019, 6, 1901547-1901555.
-

- 30. Liu, H.; Dong, J.; Zhou, H.; Yang, X.; Xu, C.; Yao, Y.; Zhou, G.; Zhang, S.; Song, Q. Real-Time Acid Rain Sensor Based on a Triboelectric Nanogenerator Made of a PTFE−PDMS Composite Film. ACS Appl. Electron. Mater. 2021, 3, 4162-4171.
-

- 31. Shima, S.; Uejima, R.; Takamura, E.; Sakamoto, H. Relationship between Output Voltage of Water Droplet-based Electricity Nanogenerator and Electrolyte Concentration. Nano Energy 2023, 112, 108503-108508.
-

- Polymer(Korea) 폴리머
- Frequency : Bimonthly(odd)
ISSN 2234-8077(Online)
Abbr. Polym. Korea - 2024 Impact Factor : 0.6
- Indexed in SCIE
 This Article
This Article
-
2025; 49(2): 253-260
Published online Mar 25, 2025
- 10.7317/pk.2025.49.2.253
- Received on Nov 1, 2024
- Revised on Nov 14, 2024
- Accepted on Nov 29, 2024
 Services
Services
- Full Text PDF
- Abstract
- ToC
- Acknowledgements
- Conflict of Interest
Introduction
Experimental
Results and Discussion
Conclusions
- References
Shared
 Correspondence to
Correspondence to
- Suk Tai Chang
-
Department of Chemical Engineering, Chung-Ang University, 84 Heukseok-ro, Dongjak-gu, Seoul 06974, Korea
- E-mail: stchang@cau.ac.kr
- ORCID:
0000-0002-1372-8872









 Copyright(c) The Polymer Society of Korea. All right reserved.
Copyright(c) The Polymer Society of Korea. All right reserved.